Simvastatin inhibits goblet cell hyperplasia and lung arginase in a mouse model of allergic asthma: a novel treatment for airway remodeling?
- PMID: 21078495
- PMCID: PMC2990975
- DOI: 10.1016/j.trsl.2010.09.003
Simvastatin inhibits goblet cell hyperplasia and lung arginase in a mouse model of allergic asthma: a novel treatment for airway remodeling?
Abstract
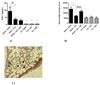
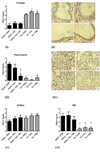
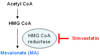
Airway remodeling in asthma contributes to airway hyperreactivity, loss of lung function, and persistent symptoms. Current therapies do not adequately treat the structural airway changes associated with asthma. The statins are cholesterol-lowering drugs that inhibit the enzyme 3-hydroxy-3-methyl-glutaryl-CoA reductase, which is the rate-limiting step of cholesterol biosynthesis in the mevalonate (MA) pathway. These drugs have been associated with improved respiratory health, and ongoing clinical trials are testing their therapeutic potential in asthma. We hypothesized that simvastatin treatment of ovalbumin (OVA)-exposed mice would attenuate early features of airway remodeling by a mevalonate-dependent mechanism. BALB/c mice initially were sensitized to OVA and then exposed to 1% OVA aerosol for 2 weeks after sensitization for 6 exposures. Simvastatin (40 mg/kg) or simvastatin plus MA (20 mg/kg) were injected intraperitoneally before each OVA exposure. Treatment with simvastatin attenuated goblet cell hyperplasia, arginase-1 protein expression, and total arginase enzyme activity, but it did not alter airway hydroxyproline content or transforming growth factor-β1. Inhibition of goblet cell hyperplasia by simvastatin was mevalonate-dependent. No appreciable changes to airway smooth muscle cells were observed in any control or treatment groups. In conclusion, in an acute mouse model of allergic asthma, simvastatin inhibited early hallmarks of airway remodeling, which are indicators that can lead to airway thickening and fibrosis. Statins are potentially novel treatments for airway remodeling in asthma. Additional studies using subchronic or chronic allergen exposure models are needed to extend these initial findings.
Copyright © 2010 Mosby, Inc. All rights reserved.
Conflict of interest statement
Figures

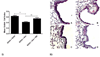




Similar articles
-
ISO-1, a macrophage migration inhibitory factor antagonist, inhibits airway remodeling in a murine model of chronic asthma.Mol Med. 2010 Sep-Oct;16(9-10):400-8. doi: 10.2119/molmed.2009.00128. Epub 2010 May 14. Mol Med. 2010. PMID: 20485865 Free PMC article.
-
Mepacrine inhibits subepithelial fibrosis by reducing the expression of arginase and TGF-beta1 in an extended subacute mouse model of allergic asthma.Am J Physiol Lung Cell Mol Physiol. 2009 Sep;297(3):L411-9. doi: 10.1152/ajplung.00138.2009. Epub 2009 Jun 19. Am J Physiol Lung Cell Mol Physiol. 2009. PMID: 19542246
-
Simvastatin inhibits airway hyperreactivity: implications for the mevalonate pathway and beyond.Am J Respir Crit Care Med. 2009 Oct 15;180(8):731-40. doi: 10.1164/rccm.200901-0018OC. Epub 2009 Jul 16. Am J Respir Crit Care Med. 2009. PMID: 19608720 Free PMC article.
-
Parabromophenacyl bromide inhibits subepithelial fibrosis by reducing TGF-β1 in a chronic mouse model of allergic asthma.Int Arch Allergy Immunol. 2015;167(2):110-8. doi: 10.1159/000434679. Epub 2015 Aug 18. Int Arch Allergy Immunol. 2015. PMID: 26303861
-
Effects of vitamin E on mitochondrial dysfunction and asthma features in an experimental allergic murine model.J Appl Physiol (1985). 2009 Oct;107(4):1285-92. doi: 10.1152/japplphysiol.00459.2009. Epub 2009 Jul 23. J Appl Physiol (1985). 2009. PMID: 19628725 Free PMC article.
Cited by
-
Innovations in asthma therapy: is there a role for inhaled statins?Expert Rev Respir Med. 2018 Jun;12(6):461-473. doi: 10.1080/17476348.2018.1457437. Epub 2018 May 3. Expert Rev Respir Med. 2018. PMID: 29575963 Free PMC article. Review.
-
Sterols and immune mechanisms in asthma.J Allergy Clin Immunol. 2023 Jan;151(1):47-59. doi: 10.1016/j.jaci.2022.09.025. Epub 2022 Oct 29. J Allergy Clin Immunol. 2023. PMID: 37138729 Free PMC article. Review.
-
A Synthetic Curcuminoid Analogue, 2,6-Bis-4-(Hydroxyl-3-Methoxybenzylidine)-Cyclohexanone (BHMC) Ameliorates Acute Airway Inflammation of Allergic Asthma in Ovalbumin-Sensitized Mice.Mediators Inflamm. 2021 Apr 3;2021:9725903. doi: 10.1155/2021/9725903. eCollection 2021. Mediators Inflamm. 2021. PMID: 33883974 Free PMC article.
-
Targeting the mevalonate cascade as a new therapeutic approach in heart disease, cancer and pulmonary disease.Pharmacol Ther. 2014 Jul;143(1):87-110. doi: 10.1016/j.pharmthera.2014.02.007. Epub 2014 Feb 26. Pharmacol Ther. 2014. PMID: 24582968 Free PMC article. Review.
-
Prophylactic benefits of systemically delivered simvastatin treatment in a house dust mite challenged murine model of allergic asthma.Br J Pharmacol. 2018 Apr;175(7):1004-1016. doi: 10.1111/bph.14140. Epub 2018 Feb 27. Br J Pharmacol. 2018. PMID: 29318574 Free PMC article.
References
-
- Vignola AM, Mirabella F, Costanzo G, et al. Airway Remodeling in Asthma. Chest. 2003;123:417S–422S. - PubMed
-
- Holgate S. Pathogenesis of Asthma. Clinical and Experimental Allergy. 2008;38:872–897. - PubMed
-
- Royce SG, Tang M. The effects of current therapies on airway remodeling in asthma and new possibilities for treatment and prevention. Curr Mol Pharmacol. 2009;2(2):169–181. - PubMed
-
- Payne DNR, Rogers AV, Adelroth E, et al. Early Thickening of the Reticular Basement Membrane in Children with Difficult Asthma. Am J Respir Crit Care Med. 2003;167:78–82. - PubMed
-
- Takeda N, Kondo M, Ito S, Ito Y, Shimokata K, Kume H. Role of RhoA inactivation in reduced cell proliferation of human airway smooth muscle by simvastatin. Am J Respir Cell Mol Biol. 2006;35(6):722–729. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

