Comparison of three ternary lipid bilayer mixtures: FRET and ESR reveal nanodomains
- PMID: 21081079
- PMCID: PMC2980711
- DOI: 10.1016/j.bpj.2010.09.064
Comparison of three ternary lipid bilayer mixtures: FRET and ESR reveal nanodomains
Abstract
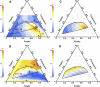
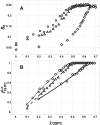
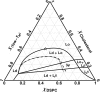
Phase diagrams of ternary lipid mixtures containing cholesterol have provided valuable insight into cell membrane behaviors, especially by describing regions of coexisting liquid-disordered (Ld) and liquid-ordered (Lo) phases. Fluorescence microscopy imaging of giant unilamellar vesicles has greatly assisted the determination of phase behavior in these systems. However, the requirement for optically resolved Ld + Lo domains can lead to the incorrect inference that in lipid-only mixtures, Ld + Lo domain coexistence generally shows macroscopic domains. Here we show this inference is incorrect for the low melting temperature phosphatidylcholines abundant in mammalian plasma membranes. By use of high compositional resolution Förster resonance energy transfer measurements, together with electron spin resonance data and spectral simulation, we find that ternary mixtures of DSPC and cholesterol together with either POPC or SOPC, do indeed have regions of Ld + Lo coexistence. However, phase domains are much smaller than the optical resolution limit, likely on the order of the Förster distance for energy transfer (R(0), ∼2-8 nm).
Copyright © 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

