Vascular pharmacology of epoxyeicosatrienoic acids
- PMID: 21081214
- PMCID: PMC3373307
- DOI: 10.1016/B978-0-12-385061-4.00002-7
Vascular pharmacology of epoxyeicosatrienoic acids
Abstract
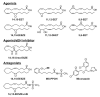
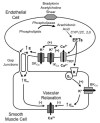
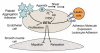
Epoxyeicosatrienoic acids (EETs) are cytochrome P450 metabolites of arachidonic acid that are produced by the vascular endothelium in responses to various stimuli such as the agonists acetylcholine (ACH) or bradykinin or by shear stress which activates phospholipase A(2) to release arachidonic acid. EETs are important regulators of vascular tone and homeostasis. In the modulation of vascular tone, EETs function as endothelium-derived hyperpolarizing factors (EDHFs). In models of vascular inflammation, EETs attenuate inflammatory signaling pathways in both the endothelium and vascular smooth muscle. Likewise, EETs regulate blood vessel formation or angiogenesis by mechanisms that are still not completely understood. Soluble epoxide hydrolase (sEH) converts EETs to dihydroxyeicosatrienoic acids (DHETs) and this metabolism limits many of the biological actions of EETs. The recent development of inhibitors of sEH provides an emerging target for pharmacological manipulation of EETs. Additionally, EETs may initiate their biological effects by interacting with a cell surface protein that is a G protein-coupled receptor (GPCR). Since GPCRs represent a common target of most drugs, further characterization of the EET receptor and synthesis of specific EET agonists and antagonist can be used to exploit many of the beneficial effects of EETs in vascular diseases, such as hypertension and atherosclerosis. This review will focus on the current understanding of the contribution of EETs to the regulation of vascular tone, inflammation, and angiogenesis. Furthermore, the therapeutic potential of targeting the EET pathway in vascular disease will be highlighted.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Epoxyeicosatrienoic acids and endothelium-dependent responses.Pflugers Arch. 2010 May;459(6):881-95. doi: 10.1007/s00424-010-0804-6. Epub 2010 Mar 12. Pflugers Arch. 2010. PMID: 20224870 Free PMC article. Review.
-
Reno-protective mechanisms of epoxyeicosatrienoic acids in cardiovascular disease.Am J Physiol Regul Integr Comp Physiol. 2012 Feb 1;302(3):R321-30. doi: 10.1152/ajpregu.00606.2011. Epub 2011 Nov 23. Am J Physiol Regul Integr Comp Physiol. 2012. PMID: 22116511 Review.
-
Epoxyeicosatrienoic acids, 20-hydroxyeicosatetraenoic acid, and renal microvascular function.Prostaglandins Other Lipid Mediat. 2013 Jul-Aug;104-105:2-7. doi: 10.1016/j.prostaglandins.2013.01.002. Epub 2013 Jan 17. Prostaglandins Other Lipid Mediat. 2013. PMID: 23333581 Free PMC article. Review.
-
Epoxyeicosatrienoic and dihydroxyeicosatrienoic acids dilate human coronary arterioles via BK(Ca) channels: implications for soluble epoxide hydrolase inhibition.Am J Physiol Heart Circ Physiol. 2006 Feb;290(2):H491-9. doi: 10.1152/ajpheart.00927.2005. Epub 2005 Oct 28. Am J Physiol Heart Circ Physiol. 2006. PMID: 16258029 Free PMC article.
-
Epoxyeicosatrienoic acid administration or soluble epoxide hydrolase inhibition attenuates renal fibrogenesis in obstructive nephropathy.Am J Physiol Renal Physiol. 2023 Feb 1;324(2):F138-F151. doi: 10.1152/ajprenal.00052.2022. Epub 2022 Dec 8. Am J Physiol Renal Physiol. 2023. PMID: 36475868 Free PMC article.
Cited by
-
Arachidonic acid monooxygenase: Genetic and biochemical approaches to physiological/pathophysiological relevance.Prostaglandins Other Lipid Mediat. 2015 Jul;120:40-9. doi: 10.1016/j.prostaglandins.2015.05.004. Epub 2015 May 15. Prostaglandins Other Lipid Mediat. 2015. PMID: 25986599 Free PMC article. Review.
-
Orally active epoxyeicosatrienoic acid analogs in hypertension and renal injury.Adv Pharmacol. 2022;94:27-55. doi: 10.1016/bs.apha.2022.02.004. Epub 2022 Mar 30. Adv Pharmacol. 2022. PMID: 35659375 Free PMC article.
-
11,12-EET increases porto-sinusoidal resistance and may play a role in endothelial dysfunction of portal hypertension.Prostaglandins Other Lipid Mediat. 2011 Nov;96(1-4):72-5. doi: 10.1016/j.prostaglandins.2011.08.002. Epub 2011 Aug 11. Prostaglandins Other Lipid Mediat. 2011. PMID: 21856435 Free PMC article.
-
Expression and Function of Eicosanoid-Producing Cytochrome P450 Enzymes in Solid Tumors.Front Pharmacol. 2020 Jun 9;11:828. doi: 10.3389/fphar.2020.00828. eCollection 2020. Front Pharmacol. 2020. PMID: 32581794 Free PMC article. Review.
-
Epoxyeicosatrienoic Acids and 20-Hydroxyeicosatetraenoic Acid on Endothelial and Vascular Function.Adv Pharmacol. 2016;77:105-41. doi: 10.1016/bs.apha.2016.04.003. Epub 2016 May 5. Adv Pharmacol. 2016. PMID: 27451096 Free PMC article. Review.
References
-
- Adeagbo ASO, Triggle CR. Varying extracellular [K]: a functional approach to separating EDHF- and EDNO-related mechanisms in perfused rat mesenteric arterial bed. J Cardiovasc Pharmacol. 1993;21:423–429. - PubMed
-
- Archer SL, Gragasin FS, Wu X, Wang S, McMurtry S, Kim DH, et al. Endothelium-derived hyperpolarizing factor in human internal mammary artery is 11,12-epoxyeicosatrienoic acid and causes relaxation by activating smooth muscle BKca channels. Circulation. 2003;107:769–776. - PubMed
-
- Bellien J, Favre J, Iacob M, Gao J, Thuillez C, Richard V, et al. Arterial stiffness is regulated by nitric oxide and endothelium-derived hyperpolarizing factor during changes in blood flow in humans. Hypertension. 2010;55:674–680. - PubMed
-
- Bellien J, Thuillez C, Joannides R. Contribution of endothelium-derived hyperpolarizing factors to the regulation of vascular tone in humans. Fundam Clin Pharmacol. 2008a;22:363–377. - PubMed
-
- Bellien J, Thuillez C, Joannides R. Role of endothelium-derived hyperpolarizing factor in the regulation of radial artery basal diameter and endothelium-dependent dilatation in vivo. Clin Exp Pharmacol Physiol. 2008b;35:494–497. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

