Nuclear activity of sperm cells during Hyacinthus orientalis L. in vitro pollen tube growth
- PMID: 21081664
- PMCID: PMC3022407
- DOI: 10.1093/jxb/erq354
Nuclear activity of sperm cells during Hyacinthus orientalis L. in vitro pollen tube growth
Abstract
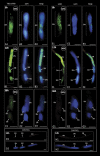
In this study, the transcriptional state and distribution of RNA polymerase II, pre-mRNA splicing machinery elements, and rRNA transcripts were investigated in the sperm cells of Hyacinthus orientalis L. during in vitro pollen tube growth. During the second pollen mitosis, no nascent transcripts were observed in the area of the dividing generative cell, whereas the splicing factors were present and their pools were divided between newly formed sperm cells. Just after their origin, the sperm cells were shown to synthesize new RNA, although at a markedly lower level than the vegetative nucleus. The occurrence of RNA synthesis was accompanied by the presence of RNA polymerase II and a rich pool of splicing machinery elements. Differences in the spatial pattern of pre-mRNA splicing factors localization reflect different levels of RNA synthesis in the vegetative nucleus and sperm nuclei. In the vegetative nucleus, they were localized homogenously, whereas in the sperm nuclei a mainly speckled pattern of small nuclear RNA with a trimethylguanosine cap (TMG snRNA) and SC35 protein distribution was observed. As pollen tube growth proceeded, inhibition of RNA synthesis in the sperm nuclei was observed, which was accompanied by a gradual elimination of the splicing factors. In addition, analysis of rRNA localization indicated that the sperm nuclei are likely to synthesize some pool of rRNA at the later steps of pollen tube. It is proposed that the described changes in the nuclear activity of H. orientalis sperm cells reflect their maturation process during pollen tube growth, and that mature sperm cells do not carry into the zygote the nascent transcripts or the splicing machinery elements.
Figures



References
-
- Bayer M, Nawy T, Giglione C, Galli M, Meinnel T, Lukowitz W. Paternal control of embryonic patterning in Arabidopsis thaliana. Science. 2009;323:1485–1488. - PubMed
-
- Bentley DL. Rules of engagement: co-transcriptional recruitment of pre-mRNA processing factors. Current Opinion in Cell Biology. 2005;17:251–256. - PubMed
-
- Berger F, Hamamura Y, Ingouff M, Higashiyama T. Double fertilization—caught in the act. Trends in Plant Science. 2008;13:437–443. - PubMed
-
- Bino RJ, Van Tuyl JM, De Vries JN. Flow cytometric determination of relative nuclear DNA contents in bicellulate and tricellulate pollen. Annals of Botany. 1990;65:3–8.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

