Immunocytochemical localization of TASK-3 (K(2P)9.1) channels in monoaminergic and cholinergic neurons
- PMID: 21082237
- PMCID: PMC11498383
- DOI: 10.1007/s10571-010-9625-6
Immunocytochemical localization of TASK-3 (K(2P)9.1) channels in monoaminergic and cholinergic neurons
Abstract
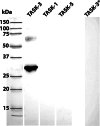
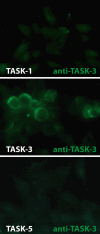
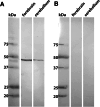
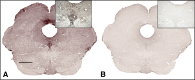
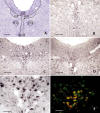
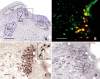
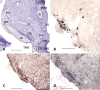
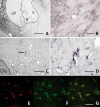
Monoaminergic and cholinergic systems are important regulators of cortical and subcortical systems, and a variety of vegetative functions are controlled by the respective neurotransmitters. Neuronal excitability and transmitter release of these neurons are strongly regulated by their potassium conductances carried by Kir and K(2P) channels. Here we describe the generation and characterization of a polyclonal monospecific antibody against rat TASK-3, a major brain K(2P) channel. After removal of cross-reactivities and affinity purification the antibody was characterized by ELISA, immunocytochemistry of TASK-3 transfected cells, and Western blots indicating that the antibody only detects TASK-3 protein, but not its paralogs TASK-1 and TASK-5. Western blot analysis of brain membrane fractions showed a single band around 45 kD, close to the predicted molecular weight of the TASK-3 protein. In addition, specific immunolabeling using the anti-TASK-3 antibody in Western blot analysis and immunocytochemistry was blocked in a concentration dependent manner by its cognate antigen only. Immunocytochemical analysis of rat brain revealed strong expression of TASK-3 channels in serotoninergic neurons of the dorsal and median raphe, noradrenergic neurons of the locus coeruleus, histaminergic neurons of the tuberomammillary nucleus and in the cholinergic neurons of the basal nucleus of Meynert. Immunofluorescence double-labeling experiments with appropriate marker enzymes confirmed the expression of TASK-3 in cholinergic, serotoninergic, and noradrenergic neurons. In the dopaminergic system strong TASK-3 expression was found in the ventral tegmental area, whereas TASK-3 immunoreactivity in the substantia nigra compacta was only weak. All immunocytochemical results were supported by in situ hybridization using TASK-3 specific riboprobes.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

