Gene expression profiling reveals Cyp26b1 to be an activin regulated gene involved in ovarian granulosa cell proliferation
- PMID: 21084447
- PMCID: PMC3033060
- DOI: 10.1210/en.2010-0749
Gene expression profiling reveals Cyp26b1 to be an activin regulated gene involved in ovarian granulosa cell proliferation
Abstract
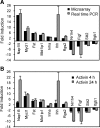
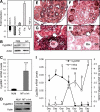
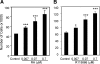
Activin, a member of the TGF-β superfamily, is an important modulator of FSH synthesis and secretion and is involved in reproductive dysfunctions and cancers. It also regulates ovarian follicle development. To understand the mechanisms and pathways by which activin regulates follicle function, we performed a microarray study and identified 240 activin regulated genes in mouse granulosa cells. The gene most strongly inhibited by activin was Cyp26b1, which encodes a P450 cytochrome enzyme that degrades retinoic acid (RA). Cyp26b1 has been shown to play an important role in male germ cell meiosis, but its expression is largely lost in the ovary around embryonic d 12.5. This study demonstrated that Cyp26b1 mRNA was expressed in granulosa cells of follicles at all postnatal developmental stages. A striking inverse spatial and temporal correlation between Cyp26b1 and activin-βA mRNA expression was observed. Cyp26b1 expression was also elevated in a transgenic mouse model that has decreased activin expression. The Cyp26 inhibitor R115866 stimulated the proliferation of primary cultured mouse granulosa cells, and a similar effect was observed with RA and activin. A pan-RA receptor inhibitor, AGN194310, abolished the stimulatory effect of either RA or activin on granulosa cell proliferation, indicating an involvement of RA receptor-mediated signaling. Overall, this study provides new insights into the mechanisms of activin action in the ovary. We conclude that Cyp26b1 is expressed in the postnatal mouse ovary, regulated by activin, and involved in the control of granulosa cell proliferation.
Figures


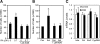
References
-
- McGee EA, Hsueh AJ 2000 Initial and cyclic recruitment of ovarian follicles. Endocr Rev 21:200–214 - PubMed
-
- Epifano O, Dean J 2002 Genetic control of early folliculogenesis in mice. Trends Endocrinol Metab 13:169–173 - PubMed
-
- Barnett KR, Schilling C, Greenfeld CR, Tomic D, Flaws JA 2006 Ovarian follicle development and transgenic mouse models. Hum Reprod Update 12:537–555 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

