Phosphorylation dynamics regulate Hsp27-mediated rescue of neuronal plasticity deficits in tau transgenic mice
- PMID: 21084594
- PMCID: PMC3073547
- DOI: 10.1523/JNEUROSCI.3155-10.2010
Phosphorylation dynamics regulate Hsp27-mediated rescue of neuronal plasticity deficits in tau transgenic mice
Abstract
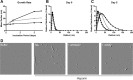
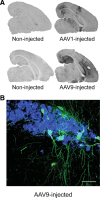
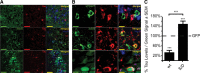
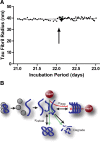
Molecular chaperones regulate the aggregation of a number of proteins that pathologically misfold and accumulate in neurodegenerative diseases. Identifying ways to manipulate these proteins in disease models is an area of intense investigation; however, the translation of these results to the mammalian brain has progressed more slowly. In this study, we investigated the ability of one of these chaperones, heat shock protein 27 (Hsp27), to modulate tau dynamics. Recombinant wild-type Hsp27 and a genetically altered version of Hsp27 that is perpetually pseudo-phosphorylated (3×S/D) were generated. Both Hsp27 variants interacted with tau, and atomic force microscopy and dynamic light scattering showed that both variants also prevented tau filament formation. However, extrinsic genetic delivery of these two Hsp27 variants to tau transgenic mice using adeno-associated viral particles showed that wild-type Hsp27 reduced neuronal tau levels, whereas 3×S/D Hsp27 was associated with increased tau levels. Moreover, rapid decay in hippocampal long-term potentiation (LTP) intrinsic to this tau transgenic model was rescued by wild-type Hsp27 overexpression but not by 3×S/D Hsp27. Because the 3×S/D Hsp27 mutant cannot cycle between phosphorylated and dephosphorylated states, we can conclude that Hsp27 must be functionally dynamic to facilitate tau clearance from the brain and rescue LTP; however, when this property is compromised, Hsp27 may actually facilitate accumulation of soluble tau intermediates.
Figures


References
-
- Barghorn S, Davies P, Mandelkow E. Tau paired helical filaments from Alzheimer's disease brain and assembled in vitro are based on beta-structure in the core domain. Biochemistry. 2004;43:1694–1703. - PubMed
-
- Berrow NS, Büssow K, Coutard B, Diprose J, Ekberg M, Folkers GE, Levy N, Lieu V, Owens RJ, Peleg Y, Pinaglia C, Quevillon-Cheruel S, Salim L, Scheich C, Vincentelli R, Busso D. Recombinant protein expression and solubility screening in Escherichia coli: a comparative study. Acta Crystallogr D Biol Crystallogr. 2006;62:1218–1226. - PubMed
-
- Bolte S, Cordelières FP. A guided tour into subcellular colocalization analysis in light microscopy. J Microsc. 2006;224:213–232. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
