Oxalyl retro-peptide gelators. Synthesis, gelation properties and stereochemical effects
- PMID: 21085503
- PMCID: PMC2981816
- DOI: 10.3762/bjoc.6.106
Oxalyl retro-peptide gelators. Synthesis, gelation properties and stereochemical effects
Abstract
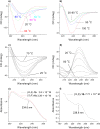
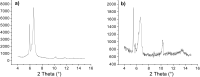
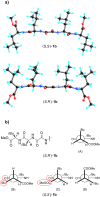
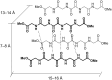
In this work we report on gelation properties, self-assembly motifs, chirality effects and morphological characteristics of gels formed by chiral retro-dipeptidic gelators in the form of terminal diacids (1a-5a) and their dimethyl ester (1b-5b) and dicarboxamide (1c-5c) derivatives. Terminal free acid retro-dipeptides (S,S)-bis(LeuLeu) 1a, (S,S)-bis(PhgPhg) 3a and (S,S)-bis(PhePhe) 5a showed moderate to excellent gelation of highly polar water/DMSO and water/DMF solvent mixtures. Retro-peptides incorporating different amino acids (S,S)-(LeuPhg) 2a and (S,S)-(PhgLeu) 4a showed no or very weak gelation. Different gelation effectiveness was found for racemic and single enantiomer gelators. The heterochiral (S,R)-1c diastereoisomer is capable of immobilizing up to 10 and 4 times larger volumes of dichloromethane/DMSO and toluene/DMSO solvent mixtures compared to homochiral (S,S)-1c. Based on the results of (1)H NMR, FTIR, CD investigations, molecular modeling and XRPD studies of diasteroisomeric diesters (S,S)-1b/(S,R)-1b and diacids (S,S)-1b/(S,R)-1a, a basic packing model in their gel aggregates is proposed. The intermolecular hydrogen bonding between extended gelator molecules utilizing both, the oxalamide and peptidic units and layered organization were identified as the most likely motifs appearing in the gel aggregates. Molecular modeling studies of (S,S)-1a/(S,R)-1a and (S,S)-1b/(S,R)-1b diasteroisomeric pairs revealed a decisive stereochemical influence yielding distinctly different low energy conformations: those of (S,R)-diastereoisomers with lipophilic i-Bu groups and polar carboxylic acid or ester groups located on the opposite sides of the oxalamide plane resembling bola amphiphilic structures and those of (S,S)-diasteroisomers possessing the same groups located at both sides of the oxalamide plane. Such conformational characteristics were found to strongly influence both, gelator effectiveness and morphological characteristics of gel aggregates.
Keywords: chiral; organogel; oxalamide; retro-peptide; self-assembly.
Figures

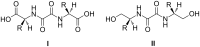



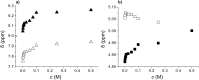
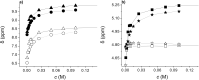
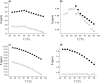
 ); racemate oxalamide-β-Leu-C*H (
); racemate oxalamide-β-Leu-C*H ( ).
).
Similar articles
-
Chiral bis(amino acid)- and bis(amino alcohol)-oxalamide gelators. Gelation properties, self-assembly motifs and chirality effects.Chem Commun (Camb). 2010 Jan 28;46(4):522-37. doi: 10.1039/b920353m. Epub 2009 Dec 3. Chem Commun (Camb). 2010. PMID: 20062853
-
Chiral hexa- and nonamethylene-bridged bis(L-Leu-oxalamide) gelators: the first oxalamide gels containing aggregates with a chiral morphology.Chemistry. 2013 Jun 24;19(26):8558-72. doi: 10.1002/chem.201300642. Epub 2013 May 7. Chemistry. 2013. PMID: 23653294
-
Chiral bis(amino alcohol)oxalamide gelators-gelation properties and supramolecular organization: racemate versus pure enantiomer gelation.Chemistry. 2003 Nov 21;9(22):5567-80. doi: 10.1002/chem.200304573. Chemistry. 2003. PMID: 14639640
-
Bis(amino acid) oxalyl amides as ambidextrous gelators of water and organic solvents: supramolecular gels with temperature dependent assembly/dissolution equilibrium.Chemistry. 2001 Aug 3;7(15):3328-41. doi: 10.1002/1521-3765(20010803)7:15<3328::aid-chem3328>3.0.co;2-c. Chemistry. 2001. PMID: 11531119
-
To gel or not to gel: correlating molecular gelation with solvent parameters.Chem Soc Rev. 2015 Oct 7;44(17):6035-58. doi: 10.1039/c5cs00136f. Epub 2015 May 5. Chem Soc Rev. 2015. PMID: 25941907 Review.
Cited by
-
Chirality-Mediated Mechanical and Structural Properties of Oligopeptide Hydrogels.Chem Mater. 2012 Jun 26;24(12):2299-2310. doi: 10.1021/cm300422q. Chem Mater. 2012. PMID: 23641124 Free PMC article.
-
Gamma Radiation- and Ultraviolet-Induced Polymerization of Bis(amino acid)fumaramide Gel Assemblies.Polymers (Basel). 2022 Jan 5;14(1):214. doi: 10.3390/polym14010214. Polymers (Basel). 2022. PMID: 35012236 Free PMC article.
-
The Metal Effect on Self-Assembling of Oxalamide Gelators Explored by Mass Spectrometry and DFT Calculations.J Am Soc Mass Spectrom. 2018 Jan;29(1):103-113. doi: 10.1007/s13361-017-1834-5. Epub 2017 Oct 30. J Am Soc Mass Spectrom. 2018. PMID: 29086338
-
Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials.Chem Rev. 2015 Dec 23;115(24):13165-307. doi: 10.1021/acs.chemrev.5b00299. Epub 2015 Dec 8. Chem Rev. 2015. PMID: 26646318 Free PMC article. Review.
-
Using chirality to influence supramolecular gelation.Chem Sci. 2019 Jul 3;10(33):7801-7806. doi: 10.1039/c9sc02239b. eCollection 2019 Sep 7. Chem Sci. 2019. PMID: 31588329 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
