Rates of intercalator-driven platination of DNA determined by a restriction enzyme cleavage inhibition assay
- PMID: 21086002
- PMCID: PMC3049956
- DOI: 10.1007/s00775-010-0733-z
Rates of intercalator-driven platination of DNA determined by a restriction enzyme cleavage inhibition assay
Abstract
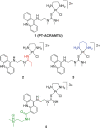
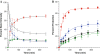
A restriction enzyme cleavage inhibition assay was designed to determine the rates of DNA platination by four non-cross-linking platinum-acridine agents represented by the formula [Pt(am(2))LCl](NO(3))(2), where am is a diamine nonleaving group and L is an acridine derived from the intercalator 1-[2-(acridin-9-ylamino)ethyl]-1,3-dimethylthiourea (ACRAMTU). The formation of monofunctional adducts in the target sequence 5'-CGA was studied in a 40-base-pair probe containing the EcoRI restriction site GAATTC. The time dependence of endonuclease inhibition was quantitatively analyzed by polyacrylamide gel electrophoresis. The formation of monoadducts is approximately 3 times faster with double-stranded DNA than with simple nucleic acid fragments. Compound 1 (am(2) is ethane-1,2-diamine, L is ACRAMTU) reacts with a first-order rate constant of k (obs) = 1.4 ± 0.37 × 10(-4) s(-1) (t (1/2) = 83 ± 22 min). Replacement of the thiourea group in ACRAMTU with an amidine group (compound 2) accelerates the rate by fourfold (k (obs) = 5.7 ± 0.58 × 10(-4) s(-1), t (1/2) = 21 ± 2 min), and introduction of a propane-1,3-diamine nonleaving group results in a 1.5-fold enhancement in reactivity (compound 3, k (obs) = 2.1 ± 0.40 × 10(-4) s(-1), t (1/2) = 55 ± 10 min) compared with the prototype. Derivative 4, containing a 4,9-disubstituted acridine threading intercalator, was the least reactive compound in the series (k (obs) = 1.1 ± 0.40 × 10(-4) s(-1), t (1/2) = 104 ± 38 min). The data suggest a correlation may exist between the binding rates and the biological activity of the compounds. Potential pharmacological advantages of rapid formation of cytotoxic monofunctional adducts over the common purine-purine cross-links are discussed.
Figures


Similar articles
-
Solution structural study of a DNA duplex containing the guanine-N7 adduct formed by a cytotoxic platinum-acridine hybrid agent.Biochemistry. 2005 Apr 26;44(16):6059-70. doi: 10.1021/bi050021b. Biochemistry. 2005. PMID: 15835895
-
Unprecedented monofunctional metalation of adenine nucleobase in guanine- and thymine-containing dinucleotide sequences by a cytotoxic platinum-acridine hybrid agent.J Am Chem Soc. 2003 Aug 13;125(32):9629-37. doi: 10.1021/ja0351443. J Am Chem Soc. 2003. PMID: 12904029
-
Metal-intercalator-mediated self-association and one-dimensional aggregation in the structure of the excised major DNA adduct of a platinum-acridine agent.J Am Chem Soc. 2004 Apr 14;126(14):4492-3. doi: 10.1021/ja038592j. J Am Chem Soc. 2004. PMID: 15070347
-
Adenine-N3 in the DNA minor groove - an emerging target for platinum containing anticancer pharmacophores.Anticancer Agents Med Chem. 2007 Jan;7(1):125-38. doi: 10.2174/187152007779313991. Anticancer Agents Med Chem. 2007. PMID: 17266509 Review.
-
Platinum-intercalator conjugates: from DNA-targeted cisplatin derivatives to adenine binding complexes as potential modulators of gene regulation.Curr Top Med Chem. 2004;4(15):1537-49. doi: 10.2174/1568026043387313. Curr Top Med Chem. 2004. PMID: 15579095 Review.
Cited by
-
PT-ACRAMTU, a platinum-acridine anticancer agent, lengthens and aggregates, but does not stiffen or soften DNA.Cell Biochem Biophys. 2013;67(3):1103-13. doi: 10.1007/s12013-013-9614-8. Cell Biochem Biophys. 2013. PMID: 23636685 Free PMC article.
-
The Next Generation of Platinum Drugs: Targeted Pt(II) Agents, Nanoparticle Delivery, and Pt(IV) Prodrugs.Chem Rev. 2016 Mar 9;116(5):3436-86. doi: 10.1021/acs.chemrev.5b00597. Epub 2016 Feb 11. Chem Rev. 2016. PMID: 26865551 Free PMC article. Review.
-
Analysis of the DNA damage produced by a platinum-acridine antitumor agent and its effects in NCI-H460 lung cancer cells.Metallomics. 2012 Jul;4(7):645-52. doi: 10.1039/c2mt20031g. Epub 2012 Mar 29. Metallomics. 2012. PMID: 22456976 Free PMC article.
-
Replacement of a thiourea with an amidine group in a monofunctional platinum-acridine antitumor agent. Effect on DNA interactions, DNA adduct recognition and repair.Mol Pharm. 2011 Oct 3;8(5):1941-54. doi: 10.1021/mp200309x. Epub 2011 Aug 17. Mol Pharm. 2011. PMID: 21806015 Free PMC article.
-
Inhibition of DNA Synthesis by a Platinum-Acridine Hybrid Agent Leads to Potent Cell Kill in Non-Small Cell Lung Cancer.ACS Med Chem Lett. 2011 Aug 31;2(11):870-874. doi: 10.1021/ml2001888. ACS Med Chem Lett. 2011. PMID: 22328962 Free PMC article.
References
-
- Kelland L. Nat Rev Cancer. 2007;7:573–584. - PubMed
-
- Cepeda V, Fuertes MA, Castilla J, Alonso C, Quevedo C, Perez JM. Anticancer Agents Med Chem. 2007;7:3–18. - PubMed
-
- van de Vaart PJ, Belderbos J, de Jong D, Sneeuw KC, Majoor D, Bartelink H, Begg AC. Int J Cancer. 2000;89:160–166. - PubMed
-
- Fujii T, Toyooka S, Ichimura K, Fujiwara Y, Hotta K, Soh J, Suehisa H, Kobayashi N, Aoe M, Yoshino T, Kiura K, Date H. Lung Cancer. 2008;59:377–384. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

