Characterization of a transgenic short hairpin RNA-induced murine model of Tafazzin deficiency
- PMID: 21091282
- PMCID: PMC3166794
- DOI: 10.1089/hum.2010.199
Characterization of a transgenic short hairpin RNA-induced murine model of Tafazzin deficiency
Abstract
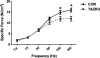
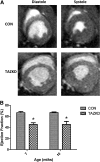
Barth's syndrome (BTHS) is an X-linked mitochondrial disease that is due to a mutation in the Tafazzin (TAZ) gene. Based on sequence homology, TAZ has been characterized as an acyltransferase involved in the metabolism of cardiolipin (CL), a unique phospholipid almost exclusively located in the mitochondrial inner membrane. Yeast, Drosophila, and zebrafish models have been invaluable in elucidating the role of TAZ in BTHS, but until recently a mammalian model to study the disease has been lacking. Based on in vitro evidence of RNA-mediated TAZ depletion, an inducible short hairpin RNA (shRNA)-mediated TAZ knockdown (TAZKD) mouse model has been developed (TaconicArtemis GmbH, Cologne, Germany), and herein we describe the assessment of this mouse line as a model of BTHS. Upon induction of the TAZ-specific shRNA in vivo, transgenic mouse TAZ mRNA levels were reduced by >89% in cardiac and skeletal muscle. TAZ deficiency led to the absence of tetralineoyl-CL and accumulation of monolyso-CL in cardiac muscle. Furthermore, mitochondrial morphology from cardiac and skeletal muscle was altered. Skeletal muscle mitochondria demonstrated disrupted cristae, and cardiac mitochondria were significantly enlarged and displace neighboring myofibrils. Physiological measurements demonstrated a reduction in isometric contractile strength of the soleus and a reduction in cardiac left ventricular ejection fraction of TAZKD mice compared with control animals. Therefore, the inducible TAZ-deficient model exhibits some of the molecular and clinical characteristics of BTHS patients and may ultimately help to improve our understanding of BTHS-related cardioskeletal myopathy as well as serve as an important tool in developing therapeutic strategies for BTHS.
Figures
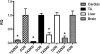


Similar articles
-
Tafazzin deficiency impairs CoA-dependent oxidative metabolism in cardiac mitochondria.J Biol Chem. 2020 Aug 28;295(35):12485-12497. doi: 10.1074/jbc.RA119.011229. Epub 2020 Jul 14. J Biol Chem. 2020. PMID: 32665401 Free PMC article.
-
Targeted overexpression of catalase to mitochondria does not prevent cardioskeletal myopathy in Barth syndrome.J Mol Cell Cardiol. 2018 Aug;121:94-102. doi: 10.1016/j.yjmcc.2018.07.001. Epub 2018 Jul 2. J Mol Cell Cardiol. 2018. PMID: 30008435 Free PMC article.
-
Barth syndrome-related cardiomyopathy is associated with a reduction in myocardial glucose oxidation.Am J Physiol Heart Circ Physiol. 2021 Jun 1;320(6):H2255-H2269. doi: 10.1152/ajpheart.00873.2020. Epub 2021 Apr 30. Am J Physiol Heart Circ Physiol. 2021. PMID: 33929899
-
Barth Syndrome: Connecting Cardiolipin to Cardiomyopathy.Lipids. 2017 Feb;52(2):99-108. doi: 10.1007/s11745-016-4229-7. Epub 2017 Jan 9. Lipids. 2017. PMID: 28070695 Free PMC article. Review.
-
Experimental models of Barth syndrome.J Inherit Metab Dis. 2022 Jan;45(1):72-81. doi: 10.1002/jimd.12423. Epub 2021 Aug 15. J Inherit Metab Dis. 2022. PMID: 34370877 Free PMC article. Review.
Cited by
-
Defective Mitochondrial Cardiolipin Remodeling Dampens HIF-1α Expression in Hypoxia.Cell Rep. 2018 Oct 16;25(3):561-570.e6. doi: 10.1016/j.celrep.2018.09.057. Cell Rep. 2018. PMID: 30332638 Free PMC article.
-
Studying Lipid-Related Pathophysiology Using the Yeast Model.Front Physiol. 2021 Oct 28;12:768411. doi: 10.3389/fphys.2021.768411. eCollection 2021. Front Physiol. 2021. PMID: 34777024 Free PMC article. Review.
-
Phospholipid ebb and flow makes mitochondria go.J Cell Biol. 2020 Aug 3;219(8):e202003131. doi: 10.1083/jcb.202003131. J Cell Biol. 2020. PMID: 32614384 Free PMC article. Review.
-
Mitochondrial dysfunctions in barth syndrome.IUBMB Life. 2019 Jul;71(7):791-801. doi: 10.1002/iub.2018. Epub 2019 Feb 11. IUBMB Life. 2019. PMID: 30746873 Free PMC article. Review.
-
Mitochondria-targeted antioxidant prevents cardiac dysfunction induced by tafazzin gene knockdown in cardiac myocytes.Oxid Med Cell Longev. 2014;2014:654198. doi: 10.1155/2014/654198. Epub 2014 Aug 27. Oxid Med Cell Longev. 2014. PMID: 25247053 Free PMC article.
References
-
- Abdul-Hussien H. Hanemaaijer R. Verheijin J.H., et al. Doxycycline therapy for abdominal aneurysm: Improved proteolytic balance through reduced neutrophil content. J. Vasc. Surg. 2009;49:741–749. - PubMed
-
- Barth P.G. Scholte H.R. Berden J.A., et al. An X-linked mitochondrial disease affecting cardiac muscle, skeletal muscle and neutrophil leucocytes. J. Neurol. Sci. 1983;62:327–355. - PubMed
-
- Barth P.G. van den Bogert C. Bolhuis P.A., et al. X-linked cardioskeletal myopathy and neutropenia (Barth syndrome): Respiratory-chain abnormalities in cultured fibroblasts. J. Inherit. Metab. Dis. 1996;19:157–160. - PubMed
-
- Barth P.G. Wanders R.J. Vreken P., et al. X-linked cardioskeletal myopathy and neutropenia (Barth syndrome) (MIM 302060) J. Inherit. Metab. Dis. 1999;22:555–567. - PubMed
-
- Barth P.G. Valianpour F. Bowen V.M., et al. X-linked cardioskeletal myopathy and neutropenia (Barth syndrome): An update. Am. J. Med. Genet. A. 2004;126A:349–354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

