Allosteric modulators induce distinct movements at the GABA-binding site interface of the GABA-A receptor
- PMID: 21093460
- PMCID: PMC3026633
- DOI: 10.1016/j.neuropharm.2010.11.009
Allosteric modulators induce distinct movements at the GABA-binding site interface of the GABA-A receptor
Abstract
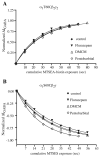
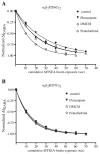
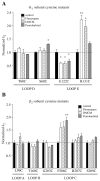
Benzodiazepines (BZDs) and barbiturates exert their CNS actions by binding to GABA-A receptors (GABARs). The structural mechanisms by which these drugs allosterically modulate GABAR function, to either enhance or inhibit GABA-gated current, are poorly understood. Here, we used the substituted cysteine accessibility method to examine and compare structural movements in the GABA-binding site interface triggered by a BZD positive (flurazepam), zero (flumazenil) and negative (3-carbomethoxy-4-ethyl-6,7-dimethoxy-β-carboline, DMCM) modulator as well as the barbiturate pentobarbital. Ten residues located throughout the GABA-binding site interface were individually mutated to cysteine. Wild-type and mutant α(1)β(2)γ(2) GABARs were expressed in Xenopus laevis oocytes and functionally characterized using two-electrode voltage clamp. We measured and compared the rates of modification of the introduced cysteines by sulfhydryl-reactive methanethiosulfonate (MTS) reagents in the absence and presence of BZD-site ligands and pentobarbital. Flurazepam and DMCM each accelerated the rate of reaction at α(1)R131C and slowed the rate of reaction at α(1)E122C, whereas flumazenil had no effect indicating that simple occupation of the BZD binding site is not sufficient to cause movements near these positions. Therefore, BZD-induced movements at these residues are likely associated with the ability of the BZD to modulate GABAR function (BZD efficacy). Low, modulating concentrations of pentobarbital accelerated the rate of reaction at α(1)S68C and β(2)P206C, slowed the rate of reaction at α(1)E122C and had no effect at α(1)R131C. These findings indicate that pentobarbital and BZDs induce different movements in the receptor, providing evidence that the structural mechanisms underlying their allosteric modulation of GABAR function are distinct.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures


Similar articles
-
Individually monitoring ligand-induced changes in the structure of the GABAA receptor at benzodiazepine binding site and non-binding-site interfaces.Mol Pharmacol. 2008 Jul;74(1):203-12. doi: 10.1124/mol.108.044891. Epub 2008 Apr 18. Mol Pharmacol. 2008. PMID: 18424553 Free PMC article.
-
A (beta)-strand in the (gamma)2 subunit lines the benzodiazepine binding site of the GABA A receptor: structural rearrangements detected during channel gating.J Neurosci. 2001 Jul 15;21(14):4977-86. doi: 10.1523/JNEUROSCI.21-14-04977.2001. J Neurosci. 2001. PMID: 11438573 Free PMC article.
-
Structural mechanisms underlying benzodiazepine modulation of the GABA(A) receptor.J Neurosci. 2008 Mar 26;28(13):3490-9. doi: 10.1523/JNEUROSCI.5727-07.2008. J Neurosci. 2008. PMID: 18367615 Free PMC article.
-
Subtype Selective γ-Aminobutyric Acid Type A Receptor (GABAAR) Modulators Acting at the Benzodiazepine Binding Site: An Update.J Med Chem. 2020 Apr 9;63(7):3425-3446. doi: 10.1021/acs.jmedchem.9b01312. Epub 2019 Dec 5. J Med Chem. 2020. PMID: 31738537 Review.
-
An updated unified pharmacophore model of the benzodiazepine binding site on gamma-aminobutyric acid(a) receptors: correlation with comparative models.Curr Med Chem. 2007;14(26):2755-75. doi: 10.2174/092986707782360097. Curr Med Chem. 2007. PMID: 18045122 Review.
Cited by
-
Regulation of the surface expression of α4β2δ GABAA receptors by high efficacy states.Brain Res. 2012 Jun 29;1463:1-20. doi: 10.1016/j.brainres.2012.04.047. Epub 2012 May 2. Brain Res. 2012. PMID: 22609410 Free PMC article.
-
Engineering a light-regulated GABAA receptor for optical control of neural inhibition.ACS Chem Biol. 2014 Jul 18;9(7):1414-9. doi: 10.1021/cb500167u. Epub 2014 May 22. ACS Chem Biol. 2014. PMID: 24819442 Free PMC article.
-
Propofol binding to the resting state of the gloeobacter violaceus ligand-gated ion channel (GLIC) induces structural changes in the inter- and intrasubunit transmembrane domain (TMD) cavities.J Biol Chem. 2013 Jun 14;288(24):17420-31. doi: 10.1074/jbc.M113.464040. Epub 2013 May 2. J Biol Chem. 2013. PMID: 23640880 Free PMC article.
-
Impaired surface αβγ GABA(A) receptor expression in familial epilepsy due to a GABRG2 frameshift mutation.Neurobiol Dis. 2013 Feb;50:135-41. doi: 10.1016/j.nbd.2012.10.008. Epub 2012 Oct 13. Neurobiol Dis. 2013. PMID: 23069679 Free PMC article.
-
Targeting GABAAR-Associated Proteins: New Modulators, Labels and Concepts.Front Mol Neurosci. 2019 Jun 26;12:162. doi: 10.3389/fnmol.2019.00162. eCollection 2019. Front Mol Neurosci. 2019. PMID: 31293385 Free PMC article.
References
-
- Amin J. A single hydrophobic residue confers barbiturate sensitivity to gamma-aminobutyric acid type C receptor. Mol Pharmacol. 1999;55:411–423. - PubMed
-
- Belelli I, Pistis I, Peters JA, Lambert JJ. General anaesthetic action at transmitter- gated inhibitory amino acid receptors. Trends Pharmacol Sci. 1999;20:496–502. - PubMed
-
- Boileau AJ, Kucken AM, Evers AR, Czajkowski C. Molecular dissection of benzodiazepine binding and allosteric coupling using chimeric gamma-aminobutyric acidA receptor subunits. Mol Pharmacol. 1998;53:295–303. - PubMed
Web References
-
- Herrington R. Controlling the False Discovery Rate in Multiple Hypothesis Testing. 2002. http://www.unt.edu/benchmarks/archives/2002/april02/rss.htm.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

