Morphological and electrical properties of oligodendrocytes in the white matter of the corpus callosum and cerebellum
- PMID: 21098009
- PMCID: PMC3055543
- DOI: 10.1113/jphysiol.2010.201376
Morphological and electrical properties of oligodendrocytes in the white matter of the corpus callosum and cerebellum
Abstract
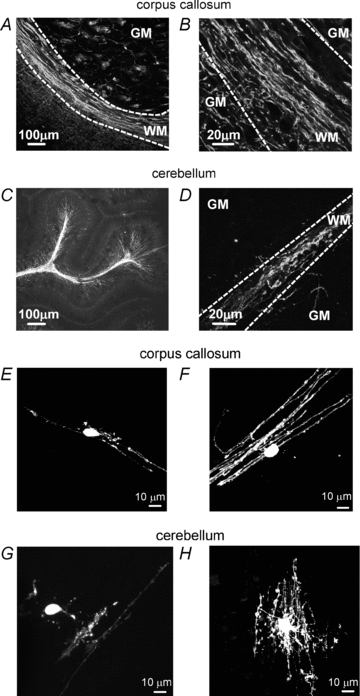
In the central nervous system, electrical signals passing along nerve cells are speeded by cells called oligodendrocytes, which wrap the nerve cells with a fatty layer called myelin. This layer is important for rapid information processing, and is often lost in disease, causing mental or physical impairment in multiple sclerosis, stroke, cerebral palsy and spinal cord injury. The myelin speeds the information flow in two ways, by decreasing the capacitance of the nerve cell and by increasing its membrane resistance, but little is known about the latter aspect of myelin function. By recording electrically from oligodendrocytes and imaging their morphology we characterised the geometry and, for the first time, the resistance of myelin in the brain. This revealed differences between the properties of oligodendrocytes in two brain areas and established that the resistance of myelin is sufficiently high to prevent significant slowing of the nerve electrical signal by current leakage through the myelin.
Figures


References
-
- Agrawal D, Hawk R, Avila RL, Inouye H, Kirschner DA. Internodal myelination during development quantitated using X-ray diffraction. J Struct Biol. 2009;168:521–526. - PubMed
-
- Alle H, Geiger JR. Combined analog and action potential coding in hippocampal mossy fibers. Science. 2006;311:1290–1293. - PubMed
-
- Baumann N, Pham-Dinh D. Biology of oligodendrocyte and myelin in the mammalian central nervous system. Physiol Rev. 2001;81:871–927. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

