A noradrenergic lesion exacerbates neurodegeneration in a Down syndrome mouse model
- PMID: 21098982
- PMCID: PMC3991557
- DOI: 10.3233/JAD-2010-101218
A noradrenergic lesion exacerbates neurodegeneration in a Down syndrome mouse model
Abstract
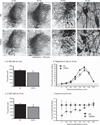
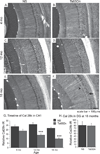
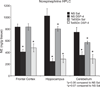
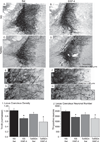
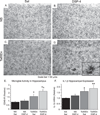
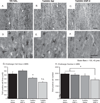
Individuals with Down syndrome (DS) acquire Alzheimer's-like dementia (AD) and associated neuropathology earlier and at significantly greater rates than age-matched normosomic individuals. However, biological mechanisms have not been discovered and there is currently limited therapy for either DS- or AD-related dementia. Segmental trisomy 16 (Ts65Dn) mice provide a useful model for many of the degenerative changes which occur with age in DS including cognitive deficits, neuroinflammation, and degeneration of basal forebrain cholinergic neurons. Loss of noradrenergic locus coeruleus (LC) neurons is an early event in AD and in DS, and may contribute to the neuropathology. We report that Ts65Dn mice exhibit progressive loss of norepinephrine (NE) phenotype in LC neurons. In order to determine whether LC degeneration contributes to memory loss and neurodegeneration in Ts65Dn mice, we administered the noradrenergic neurotoxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP-4; 2 doses of 50 mg/kg, i.p.) to Ts65Dn mice at four months of age, prior to working memory loss. At eight months of age, Ts65Dn mice treated with DSP-4 exhibited an 80% reduction in hippocampal NE, coupled with a marked increase in hippocampal neuroinflammation. Noradrenergic depletion also resulted in accelerated cholinergic neuron degeneration and a further impairment of memory function in Ts65Dn mice. In contrast, DSP-4 had minimal effects on normosomic littermates, suggesting a disease-modulated vulnerability to NE loss in the DS mouse model. These data suggest that noradrenergic degeneration may play a role in the progressive memory loss, neuroinflammation, and cholinergic loss occurring in DS individuals, providing a possible therapeutic avenue for future clinical studies.
Figures


Similar articles
-
Designer receptors enhance memory in a mouse model of Down syndrome.J Neurosci. 2015 Jan 28;35(4):1343-53. doi: 10.1523/JNEUROSCI.2658-14.2015. J Neurosci. 2015. PMID: 25632113 Free PMC article.
-
Locus Coeruleus Ablation Exacerbates Cognitive Deficits, Neuropathology, and Lethality in P301S Tau Transgenic Mice.J Neurosci. 2018 Jan 3;38(1):74-92. doi: 10.1523/JNEUROSCI.1483-17.2017. Epub 2017 Nov 13. J Neurosci. 2018. PMID: 29133432 Free PMC article.
-
Effects of long-term memantine on memory and neuropathology in Ts65Dn mice, a model for Down syndrome.Behav Brain Res. 2011 Aug 10;221(2):610-22. doi: 10.1016/j.bbr.2010.03.036. Epub 2010 Apr 2. Behav Brain Res. 2011. PMID: 20363261 Free PMC article.
-
Noradrenergic System in Down Syndrome and Alzheimer's Disease A Target for Therapy.Curr Alzheimer Res. 2016;13(1):68-83. doi: 10.2174/1567205012666150921095924. Curr Alzheimer Res. 2016. PMID: 26391048 Review.
-
Cognitive Impairment, Neuroimaging, and Alzheimer Neuropathology in Mouse Models of Down Syndrome.Curr Alzheimer Res. 2016;13(1):35-52. doi: 10.2174/1567205012666150921095505. Curr Alzheimer Res. 2016. PMID: 26391050 Free PMC article. Review.
Cited by
-
Pharmacological approaches to improving cognitive function in Down syndrome: current status and considerations.Drug Des Devel Ther. 2014 Dec 17;9:103-25. doi: 10.2147/DDDT.S51476. eCollection 2015. Drug Des Devel Ther. 2014. PMID: 25552901 Free PMC article. Review.
-
Noradrenergic dysfunction in Alzheimer's disease.Front Neurosci. 2015 Jun 17;9:220. doi: 10.3389/fnins.2015.00220. eCollection 2015. Front Neurosci. 2015. PMID: 26136654 Free PMC article. Review.
-
Metabotropic glutamate receptor 5 antagonist protects dopaminergic and noradrenergic neurons from degeneration in MPTP-treated monkeys.Brain. 2011 Jul;134(Pt 7):2057-73. doi: 10.1093/brain/awr137. Brain. 2011. PMID: 21705423 Free PMC article.
-
Rapid forgetting of social learning in the Ts65Dn mouse model of Down syndrome: New evidence for hippocampal dysfunction.Behav Neurosci. 2018 Feb;132(1):51-56. doi: 10.1037/bne0000227. Behav Neurosci. 2018. PMID: 29553775 Free PMC article.
-
Report and Research Agenda of the American Geriatrics Society and National Institute on Aging Bedside-to-Bench Conference on Sleep, Circadian Rhythms, and Aging: New Avenues for Improving Brain Health, Physical Health, and Functioning.J Am Geriatr Soc. 2016 Dec;64(12):e238-e247. doi: 10.1111/jgs.14493. Epub 2016 Nov 14. J Am Geriatr Soc. 2016. PMID: 27858974 Free PMC article.
References
-
- Mufson EJ, Ginsberg SD, Ikonomovic MD, DeKosky ST. Human cholinergic basal forebrain: chemoanatomy and neurologic dysfunction. J Chem Neuroanat. 2003;26:233–242. - PubMed
-
- Marien MR, Colpaert FC, Rosenquist AC. Noradrenergic mechanisms in neurodegenerative diseases: a theory. Brain Res Brain Res Rev. 2004;45:38–78. - PubMed
-
- Grudzien A, Shaw P, Weintraub S, Bigio E, Mash DC, Mesulam MM. Locus coeruleus neurofibrillary degeneration in aging, mild cognitive impairment and early Alzheimer’s disease. Neurobiol Aging. 2007;28:327–335. - PubMed
-
- Bondareff W, Mountjoy CQ, Roth M, Rossor MN, Iversen LL, Reynolds GP, Hauser DL. Neuronal degeneration in locus ceruleus and cortical correlates of Alzheimer disease. Alzheimer Dis Assoc Disord. 1987;1:256–262. - PubMed
-
- Abe K, Horiuchi M, Yoshimura K. Potentiation by DSP-4 of EEG slowing and memory impairment in basal forebrain-lesioned rats. Eur J Pharmacol. 1997;321:149–155. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

