Anterograde axonal transport of AAV2-GDNF in rat basal ganglia
- PMID: 21102559
- PMCID: PMC3098627
- DOI: 10.1038/mt.2010.248
Anterograde axonal transport of AAV2-GDNF in rat basal ganglia
Abstract
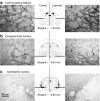
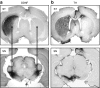
We elucidated the effects of parkinsonian degeneration on trafficking of AAV2-GDNF in the nigro-striatum (nigro-ST) of unilaterally 6-hydroxydopamine (6-OHDA)-lesioned rats. Vector infused into striatum (ST) was transported to substantia nigra (SN), both pars compacta (SNc), and pars reticulata (SNr). In the lesioned hemisphere, glial cell line-derived neurotrophic factor (GDNF) immunoreactivity was only found in SNr consistent with elimination of SNc dopaminergic (DA) neurons by 6-OHDA. Further analysis showed that striatal delivery of AAV2-GDNF resulted in GDNF expression in globus pallidus (GP), entopeduncular nucleus (EPN), and subthalamic nucleus (STN) in both lesioned and unlesioned hemispheres. Injection of vector into SN, covering both SNc and SNr, resulted in striatal expression of GDNF in the unlesioned hemisphere but not in the lesioned hemisphere. No expression was seen in GP or EPN. We conclude that adeno-associated virus serotype 2 (AAV2) is transported throughout the nigro-ST exclusively by anterograde transport. This transport phenomenon directs GDNF expression throughout the basal ganglia in regions that are adversely affected in Parkinson's disease (PD) in addition to SNc. Delivery of vector to SN, however, does not direct expression of GDNF in ST, EPN, or GP. On this basis, we believe that striatal delivery of AAV2-GDNF is the preferred course of action for trophic rescue of DA function.
Figures


Comment in
-
Moving forward toward a cure for Parkinson's: neuropathology of the nigrostriatal pathway determines the location of growth factor delivery.Mol Ther. 2011 May;19(5):827-9. doi: 10.1038/mt.2011.76. Mol Ther. 2011. PMID: 21532611 Free PMC article. No abstract available.
References
-
- Kimura M, Graybiel AM. Functions of the Cortico-basal Ganglia Loop, Springer: Tokyo; New York; 1995.
-
- Gill SS, Patel NK, Hotton GR, O'Sullivan K, McCarter R, Bunnage M, et al. Direct brain infusion of glial cell line-derived neurotrophic factor in Parkinson disease. Nat Med. 2003;9:589–595. - PubMed
-
- Lang AE, Gill S, Patel NK, Lozano A, Nutt JG, Penn R, et al. Randomized controlled trial of intraputamenal glial cell line-derived neurotrophic factor infusion in Parkinson disease. Ann Neurol. 2006;59:459–466. - PubMed
-
- Björklund A, Kirik D, Rosenblad C, Georgievska B, Lundberg C., and, Mandel RJ. Towards a neuroprotective gene therapy for Parkinson's disease: use of adenovirus, AAV and lentivirus vectors for gene transfer of GDNF to the nigrostriatal system in the rat Parkinson model. Brain Res. 2000;886:82–98. - PubMed
-
- Kordower JH. In vivo gene delivery of glial cell line–derived neurotrophic factor for Parkinson's disease. Ann Neurol. 2003;53 Suppl 3:S120–32; discussion S132. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

