3D hepatic cultures simultaneously maintain primary hepatocyte and liver sinusoidal endothelial cell phenotypes
- PMID: 21103392
- PMCID: PMC2980491
- DOI: 10.1371/journal.pone.0015456
3D hepatic cultures simultaneously maintain primary hepatocyte and liver sinusoidal endothelial cell phenotypes
Abstract
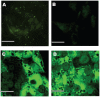
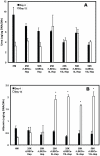
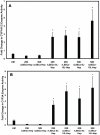
Developing in vitro engineered hepatic tissues that exhibit stable phenotype is a major challenge in the field of hepatic tissue engineering. However, the rapid dedifferentiation of hepatic parenchymal (hepatocytes) and non-parenchymal (liver sinusoidal endothelial, LSEC) cell types when removed from their natural environment in vivo remains a major obstacle. The primary goal of this study was to demonstrate that hepatic cells cultured in layered architectures could preserve or potentially enhance liver-specific behavior of both cell types. Primary rat hepatocytes and rat LSECs (rLSECs) were cultured in a layered three-dimensional (3D) configuration. The cell layers were separated by a chitosan-hyaluronic acid polyelectrolyte multilayer (PEM), which served to mimic the Space of Disse. Hepatocytes and rLSECs exhibited several key phenotypic characteristics over a twelve day culture period. Immunostaining for the sinusoidal endothelial 1 antibody (SE-1) demonstrated that rLSECs cultured in the 3D hepatic model maintained this unique feature over twelve days. In contrast, rLSECs cultured in monolayers lost their phenotype within three days. The unique stratified structure of the 3D culture resulted in enhanced heterotypic cell-cell interactions, which led to improvements in hepatocyte functions. Albumin production increased three to six fold in the rLSEC-PEM-Hepatocyte cultures. Only rLSEC-PEM-Hepatocyte cultures exhibited increasing CYP1A1/2 and CYP3A activity. Well-defined bile canaliculi were observed only in the rLSEC-PEM-Hepatocyte cultures. Together, these data suggest that rLSEC-PEM-Hepatocyte cultures are highly suitable models to monitor the transformation of toxins in the liver and their transport out of this organ. In summary, these results indicate that the layered rLSEC-PEM-hepatocyte model, which recapitulates key features of hepatic sinusoids, is a potentially powerful medium for obtaining comprehensive knowledge on liver metabolism, detoxification and signaling pathways in vitro.
Conflict of interest statement
Figures



Similar articles
-
The design of in vitro liver sinusoid mimics using chitosan-hyaluronic acid polyelectrolyte multilayers.Tissue Eng Part A. 2010 Sep;16(9):2731-41. doi: 10.1089/ten.tea.2009.0695. Tissue Eng Part A. 2010. PMID: 20491586 Free PMC article.
-
Long-term coculture strategies for primary hepatocytes and liver sinusoidal endothelial cells.Tissue Eng Part C Methods. 2015 Apr;21(4):413-22. doi: 10.1089/ten.TEC.2014.0152. Epub 2014 Nov 6. Tissue Eng Part C Methods. 2015. PMID: 25233394 Free PMC article.
-
Enhanced functional maturation of fetal porcine hepatocytes in three-dimensional poly-L-lactic acid scaffolds: a culture condition suitable for engineered liver tissues in large-scale animal studies.Cell Transplant. 2006;15(8-9):799-809. doi: 10.3727/000000006783981486. Cell Transplant. 2006. PMID: 17269450
-
Cooperation of liver cells in health and disease.Adv Anat Embryol Cell Biol. 2001;161:III-XIII, 1-151. doi: 10.1007/978-3-642-56553-3. Adv Anat Embryol Cell Biol. 2001. PMID: 11729749 Review.
-
Three-Dimensional Liver Culture Systems to Maintain Primary Hepatic Properties for Toxicological Analysis In Vitro.Int J Mol Sci. 2021 Sep 23;22(19):10214. doi: 10.3390/ijms221910214. Int J Mol Sci. 2021. PMID: 34638555 Free PMC article. Review.
Cited by
-
A genome-wide view of the de-differentiation of central nervous system endothelial cells in culture.Elife. 2020 Jan 8;9:e51276. doi: 10.7554/eLife.51276. Elife. 2020. PMID: 31913116 Free PMC article.
-
Tethered primary hepatocyte spheroids on polystyrene multi-well plates for high-throughput drug safety testing.Sci Rep. 2020 Mar 16;10(1):4768. doi: 10.1038/s41598-020-61699-4. Sci Rep. 2020. PMID: 32179810 Free PMC article.
-
Physiological oxygen tension reduces hepatocyte dedifferentiation in in vitro culture.Sci Rep. 2017 Jul 19;7(1):5923. doi: 10.1038/s41598-017-06433-3. Sci Rep. 2017. PMID: 28724942 Free PMC article.
-
Hepatocyte-like cells derived from induced pluripotent stem cells.Hepatol Int. 2017 Jan;11(1):54-69. doi: 10.1007/s12072-016-9757-y. Epub 2016 Aug 17. Hepatol Int. 2017. PMID: 27530815 Review.
-
Induced pluripotent stem cells as a source of hepatocytes.Curr Pathobiol Rep. 2014 Mar;2(1):11-20. doi: 10.1007/s40139-013-0039-2. Curr Pathobiol Rep. 2014. PMID: 25650171 Free PMC article.
References
-
- Arias IM, Boyer JL, Chisari FV, Fausto M, Schachter D, et al. The Liver: Biology and Pathology. Philadelphia, PA: Lippincott Williams and Wilkins; 2001. 1064
-
- Fraser R, Dobbs BR, Rogers GW. Lipoproteins and the liver sieve: the role of the fenestrated sinusoidal endothelium in lipoprotein metabolism, atherosclerosis, and cirrhosis. Hepatology. 1995;21:863–874. - PubMed
-
- McCuskey RS. Sinusoidal endothelial cells as an early target for hepatic toxicants. Clin Hemorheol Microcirc. 2006;34:5–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

