Inorganic nanoparticles in cancer therapy
- PMID: 21104301
- PMCID: PMC3072278
- DOI: 10.1007/s11095-010-0318-0
Inorganic nanoparticles in cancer therapy
Abstract
Nanotechnology is an evolving field with enormous potential for biomedical applications. The growing interest to use inorganic nanoparticles in medicine is due to the unique size- and shape-dependent optoelectronic properties. Herein, we will focus on gold, silver and platinum nanoparticles, discussing recent developments for therapeutic applications with regard to cancer in terms of nanoparticles being used as a delivery vehicle as well as therapeutic agents. We will also discuss some of the key challenges to be addressed in future studies.
Figures

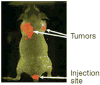

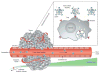

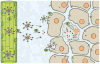
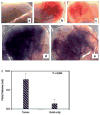


Similar articles
-
Intrinsic therapeutic applications of noble metal nanoparticles: past, present and future.Chem Soc Rev. 2012 Apr 7;41(7):2943-70. doi: 10.1039/c2cs15355f. Epub 2012 Mar 5. Chem Soc Rev. 2012. PMID: 22388295 Free PMC article. Review.
-
Biological properties of "naked" metal nanoparticles.Adv Drug Deliv Rev. 2008 Aug 17;60(11):1289-1306. doi: 10.1016/j.addr.2008.03.013. Epub 2008 Apr 10. Adv Drug Deliv Rev. 2008. PMID: 18501989 Review.
-
Gold nanoprobes for theranostics.Nanomedicine (Lond). 2011 Dec;6(10):1787-811. doi: 10.2217/nnm.11.155. Nanomedicine (Lond). 2011. PMID: 22122586 Free PMC article.
-
Strategic role of selected noble metal nanoparticles in medicine.Crit Rev Microbiol. 2016 Sep;42(5):696-719. doi: 10.3109/1040841X.2015.1018131. Epub 2015 Jun 19. Crit Rev Microbiol. 2016. PMID: 26089024 Review.
-
Inorganic hollow nanoparticles and nanotubes in nanomedicine Part 1. Drug/gene delivery applications.Drug Discov Today. 2007 Aug;12(15-16):650-6. doi: 10.1016/j.drudis.2007.06.002. Epub 2007 Jul 31. Drug Discov Today. 2007. PMID: 17706547 Review.
Cited by
-
Polyamidoamine dendrimer liposome-mediated survivin antisense oligonucleotide inhibits hepatic cancer cell proliferation by inducing apoptosis.Tumour Biol. 2014 May;35(5):5013-9. doi: 10.1007/s13277-014-1661-2. Epub 2014 Feb 3. Tumour Biol. 2014. Retraction in: Tumour Biol. 2017 Apr 20. doi: 10.1007/s13277-017-5487-6. PMID: 24488668 Retracted.
-
A facile method to prepare superparamagnetic iron oxide and hydrophobic drug-encapsulated biodegradable polyurethane nanoparticles.Int J Nanomedicine. 2017 Mar 2;12:1775-1789. doi: 10.2147/IJN.S120290. eCollection 2017. Int J Nanomedicine. 2017. PMID: 28280341 Free PMC article.
-
Biosynthesis of Gold and Silver Nanoparticles Using Phytochemical Compounds.Molecules. 2023 Apr 5;28(7):3240. doi: 10.3390/molecules28073240. Molecules. 2023. PMID: 37050004 Free PMC article. Review.
-
Noble metal nanoparticles applications in cancer.J Drug Deliv. 2012;2012:751075. doi: 10.1155/2012/751075. Epub 2011 Oct 5. J Drug Deliv. 2012. PMID: 22007307 Free PMC article.
-
Nanomechanical Insight of Pancreatic Cancer Cell Membrane during Receptor Mediated Endocytosis of Targeted Gold Nanoparticles.ACS Appl Bio Mater. 2021 Jan 18;4(1):984-994. doi: 10.1021/acsabm.0c01443. Epub 2020 Dec 30. ACS Appl Bio Mater. 2021. PMID: 34913031 Free PMC article.
References
-
- Patra CR, Bhattacharya R, Mukhopadhyay D, Mukherjee P. Application of gold nanoparticles for targeted therapy in cancer. J Biomed Nanotechnol. 2008;4:99–132.
-
- Paciotti GF, Myer L, Weinreich D, Goia D, Pavel N, McLaughlin RE, Tamarkin L. Colloidal gold: A novel nanoparticle vector for tumor directed drug delivery. Drug Deliv. 2004;11:169–183. - PubMed
-
- Mahdihassan S. Cinnabar-gold as the best alchemical drug of longevity, called Makaradhwaja in India. Am J Chin Med. 1985;13:93–108. - PubMed
-
- Danieland D, Astruc MC. Gold Nanoparticles: Assembly, Supramolecular Chemistry, Quantum-Size-Related Properties, and Applications Toward Biology, Catalysis, and Nanotechnology. Chem Rev. 2004;104:293–346. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

