Longitudinal investigation of permeability and distribution of macromolecules in mouse malignant transformation using PET
- PMID: 21106723
- PMCID: PMC3107124
- DOI: 10.1158/1078-0432.CCR-10-2049
Longitudinal investigation of permeability and distribution of macromolecules in mouse malignant transformation using PET
Abstract
Purpose: We apply positron emission tomography (PET) to elucidate changes in nanocarrier extravasation during the transition from premalignant to malignant cancer, providing insight into the use of imaging to characterize early cancerous lesions and the utility of nanoparticles in early disease.
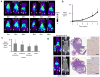
Experimental design: Albumin and liposomes were labeled with (64)Cu (half-life 12.7 hours), and longitudinal PET and CT imaging studies were conducted in a mouse model of ductal carcinoma in situ. A pharmacokinetic model was applied to estimate the tumor vascular volume and permeability.
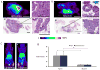
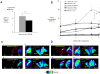
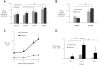
Results: From early time points characterized by disseminated hyperproliferation, the enhanced vascular permeability facilitated lesion detection. During disease progression, the vascular volume fraction increased 1.6-fold and the apparent vascular permeability to albumin and liposomes increased ∼2.5-fold to 6.6 × 10(-8) and 1.3 × 10(-8) cm/s, respectively, with the accumulation of albumin increasing earlier in the disease process. In the malignant tumor, both tracers reached similar mean intratumoral concentrations of ∼6% ID/cc but the distribution of liposomes was more heterogeneous, ranging from 1% to 18% ID/cc compared with 1% to 9% ID/cc for albumin. The tumor-to-muscle ratio was 17.9 ± 8.1 and 7.1 ± 0.5 for liposomes and albumin, respectively, indicating a more specific delivery of liposomes than with albumin.
Conclusions: PET imaging of radiolabeled particles, validated by confocal imaging and histology, detected the transition from premalignant to malignant lesions and effectively quantified the associated changes in vascular permeability.
©2010 AACR.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Figures

Similar articles
-
64Cu-Labeled tetraiodothyroacetic acid-conjugated liposomes for PET imaging of tumor angiogenesis.Nucl Med Biol. 2013 Nov;40(8):1018-24. doi: 10.1016/j.nucmedbio.2013.08.003. Epub 2013 Sep 10. Nucl Med Biol. 2013. PMID: 24035550
-
In Vivo Labeling of Serum Albumin for PET.J Nucl Med. 2014 Jul;55(7):1150-6. doi: 10.2967/jnumed.114.139642. Epub 2014 May 19. J Nucl Med. 2014. PMID: 24842890 Free PMC article.
-
Re-assessing the enhanced permeability and retention effect in peripheral arterial disease using radiolabeled long circulating nanoparticles.Biomaterials. 2016 Sep;100:101-9. doi: 10.1016/j.biomaterials.2016.05.018. Epub 2016 May 21. Biomaterials. 2016. PMID: 27254470 Free PMC article.
-
Preclinical imaging of mammary intraepithelial neoplasia with positron emission tomography.J Mammary Gland Biol Neoplasia. 2006 Apr;11(2):137-49. doi: 10.1007/s10911-006-9020-6. J Mammary Gland Biol Neoplasia. 2006. PMID: 17091397 Review.
-
The use and importance of liposomes in positron emission tomography.Drug Deliv. 2012 Jan;19(1):68-80. doi: 10.3109/10717544.2011.635721. Drug Deliv. 2012. PMID: 22211758 Review.
Cited by
-
Deciphering albumin-directed drug delivery by imaging.Adv Drug Deliv Rev. 2022 Jun;185:114237. doi: 10.1016/j.addr.2022.114237. Epub 2022 Mar 29. Adv Drug Deliv Rev. 2022. PMID: 35364124 Free PMC article. Review.
-
A physiological perspective on the use of imaging to assess the in vivo delivery of therapeutics.Ann Biomed Eng. 2014 Feb;42(2):280-98. doi: 10.1007/s10439-013-0895-2. Epub 2013 Sep 10. Ann Biomed Eng. 2014. PMID: 24018607 Free PMC article. Review.
-
Transcriptional profiling of Chinese medicinal formula Si-Wu-Tang on breast cancer cells reveals phytoestrogenic activity.BMC Complement Altern Med. 2013 Jan 10;13:11. doi: 10.1186/1472-6882-13-11. BMC Complement Altern Med. 2013. PMID: 23305139 Free PMC article.
-
Dissecting VEGF-induced acute versus chronic vascular hyperpermeability: Essential roles of dimethylarginine dimethylaminohydrolase-1.iScience. 2021 Sep 30;24(10):103189. doi: 10.1016/j.isci.2021.103189. eCollection 2021 Oct 22. iScience. 2021. PMID: 34703990 Free PMC article.
-
Ultrasonic enhancement of drug penetration in solid tumors.Front Oncol. 2013 Aug 19;3:204. doi: 10.3389/fonc.2013.00204. eCollection 2013. Front Oncol. 2013. PMID: 23967400 Free PMC article.
References
-
- Maglione JE, McGoldrick ET, Young LJ, Namba R, Gregg JP, Liu L, et al. Polyomavirus middle T-induced mammary intraepithelial neoplasia outgrowths: single origin, divergent evolution, and multiple outcomes. Mol Cancer Ther. 2004;3:941–53. - PubMed
-
- Maglione JE, Moghanaki D, Young LJ, Manner CK, Ellies LG, Joseph SO, et al. Transgenic Polyoma middle-T mice model premalignant mammary disease. Cancer Res. 2001;61:8298–305. - PubMed
-
- Abbey Craig K, Borowsky Alexander D, Gregg Jeffery P, Cardiff Robert D, Cherry Simon R. Preclinical Imaging of Mammary Intraepithelial Neoplasia with Positron Emission Tomography. J Mammary Gland Biol Neoplasia. 2006;11:137–149. - PubMed
-
- Abbey CK, Borowsky AD, McGoldrick ET, Gregg JP, Maglione JE, Cardiff RD, et al. In vivo positron-emission tomography imaging of progression and transformation in a mouse model of mammary neoplasia. Proceedings of the National Academy of Sciences of the United States of America. 2004;101:11438–43. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

