The rat kidney contains high levels of prouroguanylin (the uroguanylin precursor) but does not express GC-C (the enteric uroguanylin receptor)
- PMID: 21106860
- PMCID: PMC3280727
- DOI: 10.1152/ajprenal.00282.2010
The rat kidney contains high levels of prouroguanylin (the uroguanylin precursor) but does not express GC-C (the enteric uroguanylin receptor)
Abstract
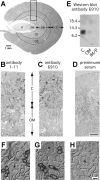
The peptide uroguanylin (Ugn) regulates enteric and renal electrolyte transport. Previous studies have shown that Ugn and its receptor GC-C (a ligand-activated guanylate cyclase) are abundant in the intestine. Less is known about Ugn and GC-C expression in the kidney. Here, we identify a 9.4-kDa polypeptide in rat kidney extracts that appears, based on its biochemical and immunological properties, to be authentic prouroguanylin (proUgn). This propeptide is relatively plentiful in the kidney (~16% of intestinal levels), whereas its mRNA is marginally present (<1% of intestinal levels), and free Ugn peptide levels are below detection limits (<0.4% of renal proUgn levels). The paucity of preproUgn-encoding mRNA and free Ugn peptide raises the possibility that the kidney might absorb intact proUgn from plasma, where the concentration of propeptide greatly exceeds that of Ugn. However, immunocytochemical analysis reveals that renal proUgn is found exclusively in distal tubular segments, sites previously shown not to accumulate radiolabeled proUgn after intravascular infusions. Thus proUgn appears to be synthesized within the kidney, but the factors that determine its abundance (rates of transcription, translation, processing, and secretion) must be balanced quite differently than in the gut. Surprisingly, we also find negligible expression of GC-C in the rat kidney, a result confirmed both by RT-PCR and by functional assays that measure Ugn-activated cGMP synthesis. Taken together, these data provide evidence for an intrarenal Ugn system that differs from the well-described intestinal system in its regulatory mechanisms and in the receptor targeted by the peptide.
Figures




Similar articles
-
Dietary salt regulates uroguanylin expression and signaling activity in the kidney, but not in the intestine.Physiol Rep. 2016 May;4(9):e12782. doi: 10.14814/phy2.12782. Epub 2016 May 15. Physiol Rep. 2016. PMID: 27185905 Free PMC article.
-
Uroguanylin, an intestinal natriuretic peptide, is delivered to the kidney as an unprocessed propeptide.Endocrinology. 2008 Sep;149(9):4486-98. doi: 10.1210/en.2007-1725. Epub 2008 May 22. Endocrinology. 2008. PMID: 18499760 Free PMC article.
-
Natriuretic and antikaliuretic effects of uroguanylin and prouroguanylin in the rat.Am J Physiol Renal Physiol. 2010 Dec;299(6):F1433-42. doi: 10.1152/ajprenal.00281.2010. Epub 2010 Sep 22. Am J Physiol Renal Physiol. 2010. PMID: 20861080 Free PMC article.
-
Cellular effects of guanylin and uroguanylin.J Am Soc Nephrol. 2006 Mar;17(3):607-16. doi: 10.1681/ASN.2005080818. Epub 2005 Dec 28. J Am Soc Nephrol. 2006. PMID: 16382016 Review.
-
Salt and water homeostasis: uroguanylin is a circulating peptide hormone with natriuretic activity.Am J Kidney Dis. 1996 Aug;28(2):296-304. doi: 10.1016/s0272-6386(96)90318-2. Am J Kidney Dis. 1996. PMID: 8768930 Review.
Cited by
-
Pendrin, a novel transcriptional target of the uroguanylin system.Cell Physiol Biochem. 2013;32(7):221-37. doi: 10.1159/000356641. Epub 2013 Dec 18. Cell Physiol Biochem. 2013. PMID: 24429828 Free PMC article. Review.
-
A uroguanylin-GUCY2C endocrine axis regulates feeding in mice.J Clin Invest. 2011 Sep;121(9):3578-88. doi: 10.1172/JCI57925. Epub 2011 Aug 25. J Clin Invest. 2011. PMID: 21865642 Free PMC article.
-
Dietary salt regulates uroguanylin expression and signaling activity in the kidney, but not in the intestine.Physiol Rep. 2016 May;4(9):e12782. doi: 10.14814/phy2.12782. Epub 2016 May 15. Physiol Rep. 2016. PMID: 27185905 Free PMC article.
-
Receptor Guanylyl Cyclase C and Cyclic GMP in Health and Disease: Perspectives and Therapeutic Opportunities.Front Endocrinol (Lausanne). 2022 Jun 29;13:911459. doi: 10.3389/fendo.2022.911459. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35846281 Free PMC article. Review.
-
Current understanding of guanylin peptides actions.ISRN Nephrol. 2013 Apr 17;2013:813648. doi: 10.5402/2013/813648. eCollection 2013. ISRN Nephrol. 2013. PMID: 24967239 Free PMC article. Review.
References
-
- Amorim JB, Musa-Aziz R, Lessa LM, Malnic G, Fonteles MC. Effect of uroguanylin on potassium and bicarbonate transport in rat renal tubules. Can J Physiol Pharmacol 84: 1003–1010, 2006 - PubMed
-
- Blanchard RK, Cousins RJ. Upregulation of rat intestinal uroguanylin mRNA by dietary zinc restriction. Am J Physiol Gastrointest Liver Physiol 272: G972–G978, 1997 - PubMed
-
- Carrithers SL, Hill MJ, Johnson BR, O'Hara SM, Jackson BA, Ott CE, Lorenz J, Mann EA, Giannella RA, Forte LR, Greenberg RN. Renal effects of uroguanylin and guanylin in vivo. Braz J Med Biol Res 32: 1337–1344, 1999 - PubMed
-
- Carrithers SL, Ott CE, Hill MJ, Johnson BR, Cai WY, Chang JJ, Shah RG, Sun CM, Mann EA, Fonteles MC, Forte LR, Jackson BA, Giannella RA, Greenberg RN. Guanylin and uroguanylin induce natriuresis in mice lacking guanylyl cyclase-C receptor. Kidney Int 65: 40–53, 2004 - PubMed
-
- Carrithers SL, Taylor B, Cai WY, Johnson BR, Ott CE, Greenberg RN, Jackson BA. Guanylyl cyclase-C receptor mRNA distribution along the rat nephron. Reg Pept 95: 65–74, 2000 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

