Impact of injury location and severity on posttraumatic epilepsy in the rat: role of frontal neocortex
- PMID: 21112931
- PMCID: PMC3116737
- DOI: 10.1093/cercor/bhq218
Impact of injury location and severity on posttraumatic epilepsy in the rat: role of frontal neocortex
Abstract
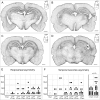
Human posttraumatic epilepsy (PTE) is highly heterogeneous, ranging from mild remitting to progressive disabling forms. PTE results in simple partial, complex partial, and secondarily generalized seizures with a wide spectrum of durations and semiologies. PTE variability is thought to depend on the heterogeneity of head injury and patient's age, gender, and genetic background. To better understand the role of these factors, we investigated the seizures resulting from calibrated fluid percussion injury (FPI) to adolescent male Sprague-Dawley rats with video electrocorticography. We show that PTE incidence and the frequency and severity of chronic seizures depend on the location and severity of FPI. The frontal neocortex was more prone to epileptogenesis than the parietal and occipital, generating earlier, longer, and more frequent partial seizures. A prominent limbic focus developed in most animals, regardless of parameters of injury. Remarkably, even with carefully controlled injury parameters, including type, severity, and location, the duration of posttraumatic apnea and the age and gender of outbred rats, there was great subject-to-subject variability in frequency, duration, and rate of progression of seizures, indicating that other factors, likely the subjects' genetic background and physiological states, have critical roles in determining the characteristics of PTE.
Figures






References
-
- Aarabi B, Taghipour M, Haghnegahdar A, Farokhi M, Mobley L. Prognostic factors in the occurrence of posttraumatic epilepsy after penetrating head injury suffered during military service. Neurosurg Focus. 2000;8(1):1–6. - PubMed
-
- Adams JH, Scott G, Parker LS, Graham DI, Doyle D. The contusion index: a quantitative approach to cerebral contusions in head injury. Neuropathol Appl Neurobiol. 1980;6:319–324. - PubMed
-
- Annegers JF, Grabow JD, Groover RV, Laws ER, Elveback LR, Kurland LT. Seizures after head trauma: a population study. Neurology. 1980;30:683–689. - PubMed
-
- Annegers JF, Hauser A, Coan SP, Rocca WA. A population-based study of seizures after traumatic brain injuries. N Engl J Med. 1998;338:20–24. - PubMed
-
- Bancaud J, Talairach J. Clinical semiology of frontal lobe seizures. Adv Neurol. 1992;57:3–58. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

