The heme sensing response regulator HssR in Staphylococcus aureus but not the homologous RR23 in Listeria monocytogenes modulates susceptibility to the antimicrobial peptide plectasin
- PMID: 21122114
- PMCID: PMC3001719
- DOI: 10.1186/1471-2180-10-307
The heme sensing response regulator HssR in Staphylococcus aureus but not the homologous RR23 in Listeria monocytogenes modulates susceptibility to the antimicrobial peptide plectasin
Abstract
Background: Host defence peptides (HDPs), also known as antimicrobial peptides (AMPs), have emerged as potential new therapeutics and their antimicrobial spectrum covers a wide range of target organisms. However, the mode of action and the genetics behind the bacterial response to HDPs is incompletely understood and such knowledge is required to evaluate their potential as antimicrobial therapeutics. Plectasin is a recently discovered HDP active against Gram-positive bacteria with the human pathogen, Staphylococcus aureus (S. aureus) being highly susceptible and the food borne pathogen, Listeria monocytogenes (L. monocytogenes) being less sensitive. In the present study we aimed to use transposon mutagenesis to determine the genetic basis for S. aureus and L. monocytogenes susceptibility to plectasin.
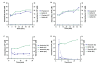
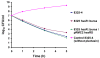
Results: In order to identify genes that provide susceptibility to plectasin we constructed bacterial transposon mutant libraries of S. aureus NCTC8325-4 and L. monocytogenes 4446 and screened for increased resistance to the peptide. No resistant mutants arose when L. monocytogenes was screened on plates containing 5 and 10 fold Minimal Inhibitory Concentration (MIC) of plectasin. However, in S. aureus, four mutants with insertion in the heme response regulator (hssR) were 2-4 fold more resistant to plectasin as compared to the wild type. The hssR mutation also enhanced resistance to the plectasin-like defensin eurocin, but not to other classes of HDPs or to other stressors tested. Addition of plectasin did not influence the expression of hssR or hrtA, a gene regulated by HssR. The genome of L. monocytogenes LO28 encodes a putative HssR homologue, RR23 (in L. monocytogenes EGD-e lmo2583) with 48% identity to the S. aureus HssR, but a mutation in the rr23 gene did not change the susceptibility of L. monocytogenes to plectasin.
Conclusions: S. aureus HssR, but not the homologue RR23 from L. monocytogenes, provides susceptibility to the defensins plectasin and eurocin. Our data suggest that a functional difference between response regulators HssR and RR23 is responsible for the difference in plectasin susceptibility observed between S. aureus and L. monocytogenes.
Figures


Similar articles
-
Antimicrobial peptides effectively kill a broad spectrum of Listeria monocytogenes and Staphylococcus aureus strains independently of origin, sub-type, or virulence factor expression.BMC Microbiol. 2008 Nov 26;8:205. doi: 10.1186/1471-2180-8-205. BMC Microbiol. 2008. PMID: 19036162 Free PMC article.
-
Signaling and DNA-binding activities of the Staphylococcus aureus HssR-HssS two-component system required for heme sensing.J Biol Chem. 2007 Sep 7;282(36):26111-21. doi: 10.1074/jbc.M703797200. Epub 2007 Jul 16. J Biol Chem. 2007. PMID: 17635909
-
'Prevalence and antimicrobial susceptibility of Listeria monocytogenes and methicillin-resistant Staphylococcus aureus strains from raw meat and meat products in Zaria, Nigeria.Lett Appl Microbiol. 2014 Mar;58(3):262-9. doi: 10.1111/lam.12183. Epub 2013 Nov 14. Lett Appl Microbiol. 2014. PMID: 24151971
-
The intrinsic cephalosporin resistome of Listeria monocytogenes in the context of stress response, gene regulation, pathogenesis and therapeutics.J Appl Microbiol. 2016 Feb;120(2):251-65. doi: 10.1111/jam.12989. Epub 2015 Dec 28. J Appl Microbiol. 2016. PMID: 26509460 Review.
-
The role of sigmaB in the stress response of Gram-positive bacteria -- targets for food preservation and safety.Curr Opin Biotechnol. 2005 Apr;16(2):218-24. doi: 10.1016/j.copbio.2005.01.008. Curr Opin Biotechnol. 2005. PMID: 15831390 Review.
Cited by
-
Importance of sigma factor mutations in increased triclosan resistance in Salmonella Typhimurium.BMC Microbiol. 2015 May 19;15:105. doi: 10.1186/s12866-015-0444-2. BMC Microbiol. 2015. PMID: 25986727 Free PMC article.
-
The antimicrobial lysine-peptoid hybrid LP5 inhibits DNA replication and induces the SOS response in Staphylococcus aureus.BMC Microbiol. 2013 Aug 14;13:192. doi: 10.1186/1471-2180-13-192. BMC Microbiol. 2013. PMID: 23945181 Free PMC article.
-
The role of ClpP, RpoS and CsrA in growth and filament formation of Salmonella enterica serovar Typhimurium at low temperature.BMC Microbiol. 2014 Aug 14;14:208. doi: 10.1186/s12866-014-0208-4. BMC Microbiol. 2014. PMID: 25123657 Free PMC article.
-
Revisiting old friends: updates on the role of two-component signaling systems in Listeria monocytogenes survival and pathogenesis.Infect Immun. 2024 Apr 9;92(4):e0034523. doi: 10.1128/iai.00345-23. Epub 2024 Mar 4. Infect Immun. 2024. PMID: 38591895 Free PMC article. Review.
References
-
- Norrby S, Nord CE, Finch R. Lack of development of new antimicrobial drugs: a potential serious threat to public health. Lancet Infect Dis. 2005;5:115–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

