Deactivation of STAT6 through serine 707 phosphorylation by JNK
- PMID: 21123173
- PMCID: PMC3030400
- DOI: 10.1074/jbc.M110.168435
Deactivation of STAT6 through serine 707 phosphorylation by JNK
Abstract
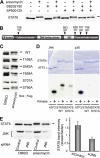
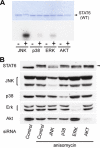
Signal transducer and activator of transcription 6 (STAT6), which plays a critical role in immune responses, is activated by interleukin-4 (IL-4). Activity of STAT family members is regulated primarily by tyrosine phosphorylations and possibly also by serine phosphorylations. Here, we report a previously undescribed serine phosphorylation of STAT6, which is activated by cell stress or by the pro-inflammatory cytokine, interleukin-1β (IL-1β). Our analyses suggest that Ser-707 is phosphorylated by c-Jun N-terminal kinase (JNK). Phosphorylation decreases the DNA binding ability of IL-4-stimulated STAT6, thereby inhibiting the transcription of STAT6-responsive genes. Inactivation of STAT6 by JNK-dependent Ser-707 phosphorylation may be one mechanism of controlling the balance between IL-1β and IL-4 signals.
Figures



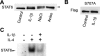

Similar articles
-
Interleukin-4-induced transcriptional activation by stat6 involves multiple serine/threonine kinase pathways and serine phosphorylation of stat6.Blood. 2000 Jan 15;95(2):494-502. Blood. 2000. PMID: 10627454
-
IL-4 induces serine phosphorylation of the STAT6 transactivation domain in B lymphocytes.Mol Immunol. 2000 Aug;37(11):641-52. doi: 10.1016/s0161-5890(00)00088-2. Mol Immunol. 2000. PMID: 11164892
-
Interleukin 4 regulates phosphorylation of serine 756 in the transactivation domain of Stat6. Roles for multiple phosphorylation sites and Stat6 function.J Biol Chem. 2004 Jun 11;279(24):25196-203. doi: 10.1074/jbc.M313668200. Epub 2004 Apr 6. J Biol Chem. 2004. PMID: 15069079
-
Signaling mechanisms, interaction partners, and target genes of STAT6.Cytokine Growth Factor Rev. 2006 Jun;17(3):173-88. doi: 10.1016/j.cytogfr.2006.01.004. Epub 2006 Mar 15. Cytokine Growth Factor Rev. 2006. PMID: 16540365 Review.
-
Transcriptional regulation by STAT6.Immunol Res. 2011 May;50(1):87-96. doi: 10.1007/s12026-011-8205-2. Immunol Res. 2011. PMID: 21442426 Free PMC article. Review.
Cited by
-
Akt, IL-4, and STAT Proteins Play Distinct Roles in Prostaglandin Production in Human Follicular Dendritic Cell-like Cells.Int J Mol Sci. 2023 Nov 24;24(23):16692. doi: 10.3390/ijms242316692. Int J Mol Sci. 2023. PMID: 38069015 Free PMC article.
-
STEAP3 promotes cancer cell proliferation by facilitating nuclear trafficking of EGFR to enhance RAC1-ERK-STAT3 signaling in hepatocellular carcinoma.Cell Death Dis. 2021 Nov 5;12(11):1052. doi: 10.1038/s41419-021-04329-9. Cell Death Dis. 2021. PMID: 34741044 Free PMC article.
-
Co-targeting JAK1/STAT6/GAS6/TAM signaling improves chemotherapy efficacy in Ewing sarcoma.Nat Commun. 2024 Jun 21;15(1):5292. doi: 10.1038/s41467-024-49667-2. Nat Commun. 2024. PMID: 38906855 Free PMC article.
-
Beneficial or Harmful Role of Macrophages in Guillain-Barré Syndrome and Experimental Autoimmune Neuritis.Mediators Inflamm. 2018 Apr 26;2018:4286364. doi: 10.1155/2018/4286364. eCollection 2018. Mediators Inflamm. 2018. PMID: 29853789 Free PMC article. Review.
-
Responses to Cytokines and Interferons that Depend upon JAKs and STATs.Cold Spring Harb Perspect Biol. 2018 Jan 2;10(1):a028555. doi: 10.1101/cshperspect.a028555. Cold Spring Harb Perspect Biol. 2018. PMID: 28620095 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

