Feedforward and recurrent inhibitory receptive fields of principal cells in the cat's dorsal lateral geniculate nucleus
- PMID: 21127903
- PMCID: PMC3023014
- DOI: 10.1007/s00424-010-0900-7
Feedforward and recurrent inhibitory receptive fields of principal cells in the cat's dorsal lateral geniculate nucleus
Abstract
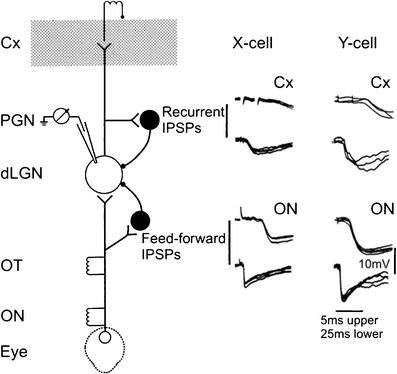
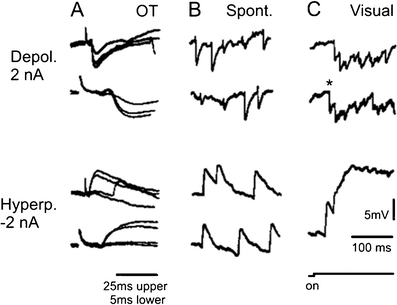
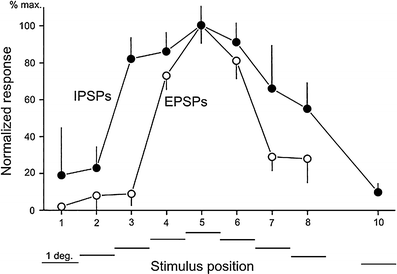
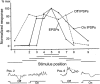
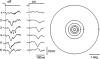
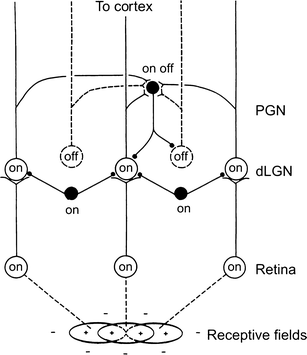
Principal cells in the dorsal lateral geniculate nucleus receive both feedforward and recurrent inhibition. Despite many years of study, the receptive field structure of these inhibitory mechanisms has not been determined. Here, we have used intracellular recordings in vivo to differentiate between the two types of inhibition and map their respective receptive fields. The feedforward inhibition of a principal cell originates from the same type of retinal ganglion cells as its excitation, while the recurrent inhibition is provided by both on- and off-centre cells. Both inhibitory effects are strongest at the centre of the excitatory receptive field. The diameter of the feedforward inhibitory field is two times larger, and the recurrent two to four times larger than the excitatory field centre. The inhibitory circuitry is similar for X and Y principal cells.
Figures



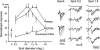
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

