Pikachurin interaction with dystroglycan is diminished by defective O-mannosyl glycosylation in congenital muscular dystrophy models and rescued by LARGE overexpression
- PMID: 21129441
- PMCID: PMC3018538
- DOI: 10.1016/j.neulet.2010.11.056
Pikachurin interaction with dystroglycan is diminished by defective O-mannosyl glycosylation in congenital muscular dystrophy models and rescued by LARGE overexpression
Abstract
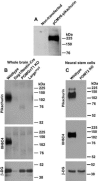
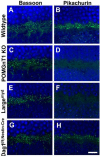
Congenital muscular dystrophies (CMD) such as muscle-eye-brain disease caused by defective glycosylation of α-dystroglycan (α-DG) exhibit defective photoreceptor synaptic function. Mouse knockouts of dystroglycan and its extracellular matrix binding partner pikachurin recapitulate this phenotype. In this study, pikachurin-α-dystroglycan interactions in several mouse models of CMD were examined by pikachurin overlay experiments. The results show that hypoglycosylation of α-dystroglycan resulted in markedly reduced pikachurin-α-dystroglycan interactions. Expression of pikachurin is abolished at the outer plexiform layer of two mouse models, protein O-mannose N-acetylglucosaminyl transferase 1 (POMGnT1) knockout and Large(myd) mice. Overexpressing LARGE restored this interaction in POMGnT1 knockout cells. These results indicate that pikachurin interactions with α-dystroglycan and its localization at the photoreceptor ribbon synapse require normal glycosylation of α-dystroglycan.
Copyright © 2010 Elsevier Ireland Ltd. All rights reserved.
Figures


References
-
- Ueda H, Gohdo T, Ohno S. Beta-dystroglycan localization in the photoreceptor and Muller cells in the rat retina revealed by immunoelectron microscopy. J. Histochem. Cytochem. 1998;46:185–191. - PubMed
-
- Schmitz F, Holbach M, Drenckhahn D. Colocalization of retinal dystrophin and actin in postsynaptic dendrites of rod and cone photoreceptor synapses. Histochemistry. 1993;100:473–479. - PubMed
-
- Cox GA, Phelps SF, Chapman VM, Chamberlain JS. New mdx mutation disrupts expression of muscle and nonmuscle isoforms of dystrophin. Nat. Genet. 1993;4:87–93. - PubMed
-
- Cibis GW, Fitzgerald KM, Harris DJ, Rothberg PG, Rupani M. The effects of dystrophin gene mutations on the ERG in mice and humans. Invest Ophthalmol. Vis. Sci. 1993;34:3646–3652. - PubMed
-
- Pillers DA, Weleber RG, Woodward WR, Green DG, Chapman VM, Ray PN. mdxCv3 mouse is a model for electroretinography of Duchenne/Becker muscular dystrophy. Invest Ophthalmol. Vis. Sci. 1995;36:462–466. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

