Mutations in two putative phosphorylation motifs in the tomato pollen receptor kinase LePRK2 show antagonistic effects on pollen tube length
- PMID: 21131355
- PMCID: PMC3039375
- DOI: 10.1074/jbc.M110.147512
Mutations in two putative phosphorylation motifs in the tomato pollen receptor kinase LePRK2 show antagonistic effects on pollen tube length
Abstract
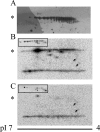
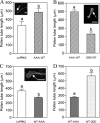
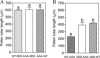
The tip-growing pollen tube is a useful model for studying polarized cell growth in plants. We previously characterized LePRK2, a pollen-specific receptor-like kinase from tomato (1). Here, we showed that LePRK2 is present as multiple phosphorylated isoforms in mature pollen membranes. Using comparative sequence analysis and phosphorylation site prediction programs, we identified two putative phosphorylation motifs in the cytoplasmic juxtamembrane (JM) domain. Site-directed mutagenesis in these motifs, followed by transient overexpression in tobacco pollen, showed that both motifs have opposite effects in regulating pollen tube length. Relative to LePRK2-eGFP pollen tubes, alanine substitutions in residues of motif I, Ser(277)/Ser(279)/Ser(282), resulted in longer pollen tubes, but alanine substitutions in motif II, Ser(304)/Ser(307)/Thr(308), resulted in shorter tubes. In contrast, phosphomimicking aspartic substitutions at these residues gave reciprocal results, that is, shorter tubes with mutations in motif I and longer tubes with mutations in motif II. We conclude that the length of pollen tubes can be negatively and positively regulated by phosphorylation of residues in motif I and II respectively. We also showed that LePRK2-eGFP significantly decreased pollen tube length and increased pollen tube tip width, relative to eGFP tubes. The kinase activity of LePRK2 was relevant for this phenotype because tubes that expressed a mutation in a lysine essential for kinase activity showed the same length and width as the eGFP control. Taken together, these results suggest that LePRK2 may have a central role in pollen tube growth through regulation of its own phosphorylation status.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

