Synthesis of 6-amino-5-cyano-1,4-disubstituted-2(1H)-pyrimidinones via copper-(I)-catalyzed alkyne-azide 'click chemistry' and their reactivity
- PMID: 21131903
- PMCID: PMC6259112
- DOI: 10.3390/molecules1512884
Synthesis of 6-amino-5-cyano-1,4-disubstituted-2(1H)-pyrimidinones via copper-(I)-catalyzed alkyne-azide 'click chemistry' and their reactivity
Abstract
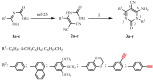
In this paper we present the room temperature synthesis of a novel serie of 1,4-disubstituted-1,2,3-triazoles 4a-l by employing the (3+2) cycloaddition reaction of pyrimidinones containing alkyne functions with different model azides in the presence of copper sulphate and sodium ascorbate. To obtain the final triazoles, we also synthesized the major precursors 6-amino-5-cyano-1,4-disubstituted-2(1H)-pyrimidinones 3a-r from ethyl 2,2-dicyanovinylcarbamate derivatives 2a-c and various primary aromatic amines containing an alkyne group. The triazoles were prepared in good to very good yields.
Figures
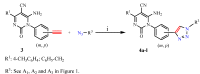

References
-
- Yi L., Shi J., Gao S., Li S., Niu C., Xi Z. Sulfonium alkylation followed by ‘click’ chemistry for facile surface modification of proteins and tobacco mosaic virus. Tetrahedron Lett. 2009;50:759–762. doi: 10.1016/j.tetlet.2008.11.107. - DOI
-
- Bock V.D., Hiemstra H., van Maarseveen J.H. CuI-Catalyzed Alkyne-Azide“Click”Cycloadditions from a Mechanistic and Synthetic Perspective. Eur. J. Org. Chem. 2006;1:51–68.
-
- Fournier D., De Geest B.G., Du Prez F.E. On-demand click functionalization of polyurethane films and foams. Polymer. 2009;50:5362–5367. doi: 10.1016/j.polymer.2009.09.047. - DOI
-
- Fan W.Q., Katritzky A.R. 1,2,3-Triazoles. Compr. Heterocycl. Chem. II. 1996;4:1–126.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

