Retinoid X receptor gamma signaling accelerates CNS remyelination
- PMID: 21131950
- PMCID: PMC4013508
- DOI: 10.1038/nn.2702
Retinoid X receptor gamma signaling accelerates CNS remyelination
Abstract
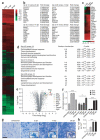
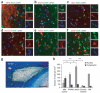
The molecular basis of CNS myelin regeneration (remyelination) is poorly understood. We generated a comprehensive transcriptional profile of the separate stages of spontaneous remyelination that follow focal demyelination in the rat CNS and found that transcripts that encode the retinoid acid receptor RXR-γ were differentially expressed during remyelination. Cells of the oligodendrocyte lineage expressed RXR-γ in rat tissues that were undergoing remyelination and in active and remyelinated multiple sclerosis lesions. Knockdown of RXR-γ by RNA interference or RXR-specific antagonists severely inhibited oligodendrocyte differentiation in culture. In mice that lacked RXR-γ, adult oligodendrocyte precursor cells efficiently repopulated lesions after demyelination, but showed delayed differentiation into mature oligodendrocytes. Administration of the RXR agonist 9-cis-retinoic acid to demyelinated cerebellar slice cultures and to aged rats after demyelination caused an increase in remyelinated axons. Our results indicate that RXR-γ is a positive regulator of endogenous oligodendrocyte precursor cell differentiation and remyelination and might be a pharmacological target for regenerative therapy in the CNS.
Figures




Comment in
-
An RXR-γ Rx for white-matter damage.Nat Neurosci. 2011 Jan;14(1):3-5. doi: 10.1038/nn0111-3. Nat Neurosci. 2011. PMID: 21187846 No abstract available.
References
-
- Prineas JW, Connell F. Remyelination in multiple sclerosis. Ann. Neurol. 1979;5:22–31. - PubMed
-
- Patani R, Balaratnam M, Vora A, Reynolds R. Remyelination can be extensive in multiple sclerosis despite a long disease course. Neuropathol. Appl. Neurobiol. 2007;33:277–287. - PubMed
-
- Raine CS, Wu E. Multiple sclerosis: remyelination in acute lesions. J. Neuropathol. Exp. Neurol. 1993;52:199–204. - PubMed
-
- Goldschmidt T, Antel J, König FB, Brück W, Kuhlmann T. Remyelination capacity of the MS brain decreases with disease chronicity. Neurology. 2009;72:1914–1921. - PubMed
-
- Franklin RJM. Why does remyelination fail in multiple sclerosis? Nat. Rev. Neurosci. 2002;3:705–714. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

