GNL3L depletion destabilizes MDM2 and induces p53-dependent G2/M arrest
- PMID: 21132010
- PMCID: PMC3072457
- DOI: 10.1038/onc.2010.550
GNL3L depletion destabilizes MDM2 and induces p53-dependent G2/M arrest
Abstract
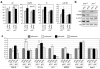
Guanine nucleotide binding protein-like 3-like (GNL3L) is a nucleolar protein and the vertebrate paralogue of nucleostemin (NS). We previously reported that nucleoplasmic mobilization of NS stabilizes MDM2 (mouse double minute 2). Here, we investigated the role of GNL3L as a novel MDM2 regulator. We found that GNL3L binds MDM2 in vivo and displays the same function as NS in stabilizing MDM2 protein and preventing its ubiquitylation. The interaction between GNL3L and MDM2 also takes place in the nucleoplasm. However, the MDM2 regulatory activity of GNL3L occurs constitutively and does not so much depend on the nucleolar release mechanism as NS does. GNL3L depletion triggers G2/M arrest in the p53-wild-type HCT116 cells more than in the p53-null cells, and upregulates specific p53 targets (that is, Bax, 14-3-3σ and p21) without affecting the ubiquitylation or stability of p53 proteins. The inhibitory activity of GNL3L on p53-mediated transcription correlates with the increased expression of GNL3L and reduced expression of 14-3-3σ and p21 in human gastrointestinal tumors. This work shows that in contrast to most nucleolar proteins that negatively control MDM2, GNL3L and NS are the only two that are designed to stabilize MDM2 protein under basal or induced condition, respectively, and may act as tumor-promoting genes.
Figures




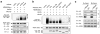

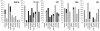
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

