Self-aligning amelogenin nanoribbons in oil-water system
- PMID: 21134461
- PMCID: PMC3204882
- DOI: 10.1016/j.jsb.2010.11.027
Self-aligning amelogenin nanoribbons in oil-water system
Abstract
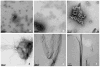
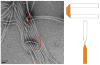
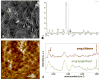
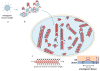
The highly organized microstructure of dental enamel is a result of protein-guided anisotropic growth of apatite nanofibers. It is established that amelogenin proteins, the main constituent of the developing enamel matrix, form nanospheres in vitro, but the amphiphilic nature of the full-length protein conveys the possibility of generating more complex structures as observed with other surfactant-like molecules. This study tested if the use of metastable oil-water emulsions can induce supramolecular assemblies of amelogenin. Recombinant full-length amelogenin, rH174, was mixed into octanol/ethyl acetate preparations of different ratios to form emulsions at pH 4.5 and 7.4. Atomic force and electron microscopy showed the formation of 16.7±1.0nm wide nanoribbons which grew to several micrometer length over a period of days. Nanoribbons formed from reverse micelles by enabling hydrophobic tails of the molecules to interact while preventing the formation of amelogenin nanospheres. Ribbon formation required the presence of calcium and phosphate ions and may be localized at a dark central line along the amelogenin ribbons. The ribbons have a strong tendency to align in parallel maintaining 5-20nm space between each other. The growth rates and number of ribbons were significantly higher at pH 4.5 and related to the metastability of the emulsion. A model for ribbon extension proposes the addition of short segments or amelogenin dimers to the ends of the ribbon. The formation of self-aligning and uniaxially elongating amelogenin structures triggered by the presence of calcium and phosphate may represent a suitable new model for protein controlled mineralization in enamel.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures




References
-
- Aichmayer B, Margolis HC, Sigel R, Yamakoshi Y, Simmer JP, Fratzl P. The onset of amelogenin nanosphere aggregation studied by small-angle X-ray scattering and dynamic light scattering. J Struct Biol. 2005;151:239–49. - PubMed
-
- Beniash E, Simmer JP, Margolis HC. The effect of recombinant mouse amelogenins on the formation and organization of hydroxyapatite crystals in vitro. J Struct Biol. 2005;149:182–90. - PubMed
-
- Berne BJ, Pecora R. Laser Light-Scattering from Liquids. Annual Review of Physical Chemistry. 1974;25:233–253.
-
- Chen Y, Wu QS, Dingibi Y. Stepwise assembly of nanoparticies, -tubes, -rods, and -wires in reverse micelle systems. European Journal of Inorganic Chemistry. 2007:4906–4910.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

