A novel RET inhibitor with potent efficacy against medullary thyroid cancer in vivo
- PMID: 21134556
- PMCID: PMC3088305
- DOI: 10.1016/j.surg.2010.09.026
A novel RET inhibitor with potent efficacy against medullary thyroid cancer in vivo
Abstract
Background: Most medullary thyroid carcinomas (MTC) recur or progress despite curative resection. Current targeted therapies show promise but lack durable efficacy and tolerability. The purpose of this study was to build on previous in vitro work and evaluate withaferin A (WA), a novel RET inhibitor, in a metastatic murine model of MTC.
Methods: A total of 5 million DRO-81-1 human MTC cells injected in the left posterior neck of nu/nu mice generated metastases uniformly to the liver, spleen, and/or lungs. Treatment with WA (8 mg/kg/day, intraperitoneally, for 21 days) was started for neoplasms > 100 mm(3). Endpoints were survival, neoplasm > 15,00 mm(3), decreased body weight, or body score (all measured three times/wk).
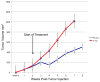
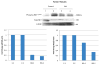
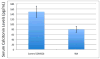
Results: All controls (saline; n = 5) died or deteriorated from metastatic disease by 7 weeks postinjection. All treated animals were alive (WA; n = 5), having tumor regression and growth delay without toxicity or weight loss at 6 weeks posttreatment (P < .01). Tumor cells treated with WA demonstrated inhibition of total and phospho-RET levels by Western blot analysis in a dose-dependent manner (almost complete inhibition with treatment of 5 μM WA) as well as potent inhibition of phospho-ERK and phospho-Akt levels.
Conclusion: WA is a novel natural-product RET-inhibitor with efficacy in a metastatic murine model of MTC. Further long-term efficacy/toxicity studies are warranted to evaluate this compound for clinical translation.
Copyright © 2010 Mosby, Inc. All rights reserved.
Figures





Similar articles
-
Novel withanolides target medullary thyroid cancer through inhibition of both RET phosphorylation and the mammalian target of rapamycin pathway.Surgery. 2012 Dec;152(6):1238-47. doi: 10.1016/j.surg.2012.08.031. Surgery. 2012. PMID: 23158190 Free PMC article.
-
A transplantable human medullary thyroid carcinoma as a model for RET tyrosine kinase-driven tumorigenesis.Endocr Relat Cancer. 2007 Jun;14(2):433-44. doi: 10.1677/ERC-06-0033. Endocr Relat Cancer. 2007. PMID: 17639056
-
In vitro and in vivo activity of cabozantinib (XL184), an inhibitor of RET, MET, and VEGFR2, in a model of medullary thyroid cancer.Thyroid. 2013 Dec;23(12):1569-77. doi: 10.1089/thy.2013.0137. Epub 2013 Sep 17. Thyroid. 2013. PMID: 23705946 Free PMC article.
-
Molecular genetics, therapeutics and RET inhibitor resistance for medullary thyroid carcinoma and future perspectives.Cell Commun Signal. 2024 Sep 28;22(1):460. doi: 10.1186/s12964-024-01837-x. Cell Commun Signal. 2024. PMID: 39342195 Free PMC article. Review.
-
Systemic Therapies for Advanced Medullary Thyroid Carcinoma.Recent Results Cancer Res. 2025;223:293-307. doi: 10.1007/978-3-031-80396-3_12. Recent Results Cancer Res. 2025. PMID: 40102263 Review.
Cited by
-
RET TKI: potential role in thyroid cancers.Curr Oncol Rep. 2012 Apr;14(2):97-104. doi: 10.1007/s11912-012-0217-0. Curr Oncol Rep. 2012. PMID: 22286373 Review.
-
Novel natural withanolides induce apoptosis and inhibit migration of neuroblastoma cells through down regulation of N-myc and suppression of Akt/mTOR/NF-κB activation.Oncotarget. 2018 Feb 7;9(18):14509-14523. doi: 10.18632/oncotarget.24429. eCollection 2018 Mar 6. Oncotarget. 2018. PMID: 29581860 Free PMC article.
-
Withaferin a alone and in combination with cisplatin suppresses growth and metastasis of ovarian cancer by targeting putative cancer stem cells.PLoS One. 2014 Sep 29;9(9):e107596. doi: 10.1371/journal.pone.0107596. eCollection 2014. PLoS One. 2014. PMID: 25264898 Free PMC article.
-
The Therapeutic Effects of Withaferin A against Cancer: Overview and Updates.Curr Mol Med. 2024;24(4):404-418. doi: 10.2174/1566524023666230418094708. Curr Mol Med. 2024. PMID: 37076466 Review.
-
Withania somnifera: Progress towards a Pharmaceutical Agent for Immunomodulation and Cancer Therapeutics.Pharmaceutics. 2022 Mar 10;14(3):611. doi: 10.3390/pharmaceutics14030611. Pharmaceutics. 2022. PMID: 35335986 Free PMC article. Review.
References
-
- O’Riordain DS, O’Brien T, Weaver AL, et al. Medullary thyroid carcinoma in multiple endocrine neoplasia types 2A and 2B. Surgery. 1994;116:1017–23. - PubMed
-
- Cohen MS, Moley JF. Surgical Treatment of Medullary Thyroid Carcinoma. Journal of Internal Medicine. 2003;253:1–11. - PubMed
-
- Brierley J, Tsang R, Simpson WJ, et al. Medullary thyroid cancer: analyses of survival and prognostic factors and the role of radiation therapy in local control. Thyroid. 1996;6:305–10. - PubMed
-
- Tisell LE, Dilley WG, Wells SA., Jr Progression of postoperative residual medullary thyroid carcinoma as monitored by plasma calcitonin levels. Surgery. 1996;119:34–9. - PubMed
-
- Martins RG, Rajendran JG, Capell P, et al. Medullary Thyroid Cancer: Options for Systemic Therapy of Metastatic Disease? J Clin Oncol. 2006;24(11):1653–1655. - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

