Antifungal mechanisms by which a novel Pseudomonas aeruginosa phenazine toxin kills Candida albicans in biofilms
- PMID: 21143312
- PMCID: PMC3828654
- DOI: 10.1111/j.1365-2958.2010.07414.x
Antifungal mechanisms by which a novel Pseudomonas aeruginosa phenazine toxin kills Candida albicans in biofilms
Abstract
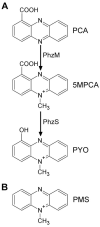
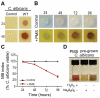
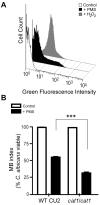
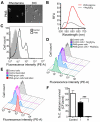
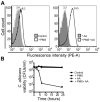
Pseudomonas aeruginosa produces several phenazines including the recently described 5-methyl-phenazine-1-carboxylic acid (5MPCA), which exhibits a novel antibiotic activity towards pathogenic fungi such as Candida albicans. Here we characterize the unique antifungal mechanisms of 5MPCA using its analogue phenazine methosulphate (PMS). Like 5MPCA, PMS induced fungal red pigmentation and killing. Mass spectrometry analyses demonstrated that PMS can be covalently modified by amino acids, a process that yields red derivatives. Furthermore, soluble proteins from C. albicans grown with either PMS or P. aeruginosa were also red and demonstrated absorbance and fluorescence spectra similar to that of PMS covalently linked to either amino acids or proteins in vitro, suggesting that 5MPCA modification by protein amine groups occurs in vivo. The red-pigmented C. albicans soluble proteins were reduced by NADH and spontaneously oxidized by oxygen, a reaction that likely generates reactive oxygen species (ROS). Additional evidence indicated that ROS generation precedes 5MPCA-induced fungal death. Reducing conditions greatly enhanced PMS uptake by C. albicans and killing. Since 5MPCA was more toxic than other phenazines that are not modified, such as pyocyanin, we propose that the covalent binding of 5MPCA promotes its accumulation in target cells and contributes to its antifungal activity in mixed-species biofilms.
© 2010 Blackwell Publishing Ltd.
Figures

References
-
- Anjaiah V, Koedam N, Nowrk-Thompson B, Loper J, Höfte M, Tambong J, Cornelis P. Involvement of phenazines and anthranilate in the antagonism of Pseudomonas aeruginosa PNA1 and Tn5 derivatives Toward Fusarium spp. and Pythium spp. Molecular Plant-Microbe interactions. 1998;11:847–854.
-
- Barelle CJ, Manson CL, MacCallum DM, Odds FC, Gow NA, Brown AJ. GFP as a quantitative reporter of gene regulation in Candida albicans. Yeast. 2004;21:333–340. - PubMed
-
- Bauernfeind A, Bertele RM, Harms K, Horl G, Jungwirth R, Petermuller C, Przyklenk B, Weisslein-Pfister C. Qualitative and quantitative microbiological analysis of sputa of 102 patients with cystic fibrosis. Infection. 1987;15:270–277. - PubMed
-
- Bisschop A, Bergsma J, Konings WN. Site of interaction between phenazine methosulphate and the respiratory chain of Bacillus subtilis. Eur J Biochem. 1979;93:369–374. - PubMed

