Immunity status of adults and children against poliomyelitis virus type 1 strains CHAT and Sabin (LSc-2ab) in Germany
- PMID: 21143885
- PMCID: PMC3022830
- DOI: 10.1186/1471-2334-10-347
Immunity status of adults and children against poliomyelitis virus type 1 strains CHAT and Sabin (LSc-2ab) in Germany
Abstract
Background: In October 2007, the working group CEN/TC 216 of the European Committee for standardisation suggested that the Sabin oral poliovirus vaccine type 1 strain (LSc-2ab) presently used for virucidal tests should be replaced by another attenuated vaccine poliovirus type 1 strain, CHAT. Both strains were historically used as oral vaccines, but the Sabin type 1 strain was acknowledged to be more attenuated. In Germany, vaccination against poliomyelitis was introduced in 1962 using the oral polio vaccine (OPV) containing Sabin strain LSc-2ab. The vaccination schedule was changed from OPV to an inactivated polio vaccine (IPV) containing wild polio virus type 1 strain Mahoney in 1998. In the present study, we assessed potential differences in neutralising antibody titres to Sabin and CHAT in persons with a history of either OPV, IPV, or OPV with IPV booster.
Methods: Neutralisation poliovirus antibodies against CHAT and Sabin 1 were measured in sera of 41 adults vaccinated with OPV. Additionally, sera from 28 children less than 10 years of age and immunised with IPV only were analysed. The neutralisation assay against poliovirus was performed according to WHO guidelines.
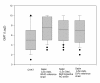
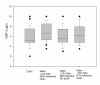
Results: The neutralisation activity against CHAT in adults with OPV vaccination history was significantly lower than against Sabin poliovirus type 1 strains (Wilcoxon signed-rank test P < 0.025). In eight sera, the antibody titres measured against CHAT were less than 8, although the titre against Sabin 1 varied between 8 and 64. Following IPV booster, anti-CHAT antibodies increased rapidly in sera of CHAT-negative adults with OPV history. Sera from children with IPV history neutralised CHAT and Sabin 1 strains equally.
Conclusion: The lack of neutralising antibodies against the CHAT strain in persons vaccinated with OPV might be associated with an increased risk of reinfection with the CHAT polio virus type 1, and this implies a putative risk of transmission of the virus to polio-free communities. We strongly suggest that laboratory workers who were immunised with OPV receive a booster vaccination with IPV before handling CHAT in the laboratory.
Figures
Similar articles
-
Surveillance of immunity acquired from poliovirus immunization including vaccination with the Sabin strain-derived inactivated vaccine.Hum Vaccin Immunother. 2019;15(5):1154-1159. doi: 10.1080/21645515.2019.1572408. Epub 2019 Mar 11. Hum Vaccin Immunother. 2019. PMID: 30676843 Free PMC article.
-
Vaccine-induced mucosal immunity to poliovirus: analysis of cohorts from an open-label, randomised controlled trial in Latin American infants.Lancet Infect Dis. 2016 Dec;16(12):1377-1384. doi: 10.1016/S1473-3099(16)30169-4. Epub 2016 Sep 13. Lancet Infect Dis. 2016. PMID: 27638357 Free PMC article. Clinical Trial.
-
Immune Serum From Sabin Inactivated Poliovirus Vaccine Immunization Neutralizes Multiple Individual Wild and Vaccine-Derived Polioviruses.Clin Infect Dis. 2017 May 15;64(10):1317-1325. doi: 10.1093/cid/cix110. Clin Infect Dis. 2017. PMID: 28419204 Clinical Trial.
-
Development of inactivated poliovirus vaccine from Sabin strains: A progress report.Biologicals. 2016 Nov;44(6):581-587. doi: 10.1016/j.biologicals.2016.08.005. Epub 2016 Oct 5. Biologicals. 2016. PMID: 27720268 Review.
-
Risks associated with the use of live-attenuated vaccine poliovirus strains and the strategies for control and eradication of paralytic poliomyelitis.Expert Rev Vaccines. 2012 May;11(5):609-28. doi: 10.1586/erv.12.28. Expert Rev Vaccines. 2012. PMID: 22827246 Review.
Cited by
-
Development of a poliovirus neutralization test with poliovirus pseudovirus for measurement of neutralizing antibody titer in human serum.Clin Vaccine Immunol. 2011 Nov;18(11):1889-94. doi: 10.1128/CVI.05225-11. Epub 2011 Aug 31. Clin Vaccine Immunol. 2011. PMID: 21880850 Free PMC article.
-
A phase I randomized clinical trial of candidate human immunodeficiency virus type 1 vaccine MVA.HIVA administered to Gambian infants.PLoS One. 2013 Oct 24;8(10):e78289. doi: 10.1371/journal.pone.0078289. eCollection 2013. PLoS One. 2013. PMID: 24205185 Free PMC article. Clinical Trial.
-
Evaluation of a virucidal quantitative carrier test for surface disinfectants.PLoS One. 2014 Jan 27;9(1):e86128. doi: 10.1371/journal.pone.0086128. eCollection 2014. PLoS One. 2014. PMID: 24475079 Free PMC article.
-
PedVacc 002: a phase I/II randomized clinical trial of MVA.HIVA vaccine administered to infants born to human immunodeficiency virus type 1-positive mothers in Nairobi.Vaccine. 2014 Oct 7;32(44):5801-8. doi: 10.1016/j.vaccine.2014.08.034. Epub 2014 Aug 27. Vaccine. 2014. PMID: 25173484 Free PMC article. Clinical Trial.
References
-
- EN 14476:2007-02: Chemical disinfectants and antiseptics - Virucidal quantitative suspensiontest for chemical disinfectants and antiseptics used in human medicine - Test method and requirements (phase 2/step 1) 2007.
-
- Cabasso VJ, Jervis G, Moyer AW, Roca-Garcia M, Orsi EV, Cox HR. Cumulative testing experience with consecutive lots of oral poliomyelitis vaccine. p. 102-134. In Live poliovirus vaccines. First International Conference on Live Poliovirus Vaccines. Scientific publication no. 44. Pan American Sanitary Bureau. Washington. D.C. 1959.
-
- Sabin AB, Boulger LR. History of Sabin attenuated poliovirus oral live vaccines. J Biol Stand. 1973;1:115–118. doi: 10.1016/0092-1157(73)90048-6. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

