TRPM7 regulates gastrulation during vertebrate embryogenesis
- PMID: 21145885
- PMCID: PMC3292586
- DOI: 10.1016/j.ydbio.2010.11.034
TRPM7 regulates gastrulation during vertebrate embryogenesis
Abstract
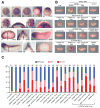
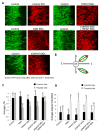
During gastrulation, cells in the dorsal marginal zone polarize, elongate, align and intercalate to establish the physical body axis of the developing embryo. Here we demonstrate that the bifunctional channel-kinase TRPM7 is specifically required for vertebrate gastrulation. TRPM7 is temporally expressed maternally and throughout development, and is spatially enriched in tissues undergoing convergent extension during gastrulation. Functional studies reveal that TRPM7's ion channel, but not its kinase domain, specifically affects cell polarity and convergent extension movements during gastrulation, independent of mesodermal specification. During gastrulation, the non-canonical Wnt pathway via Dishevelled (Dvl) orchestrates the activities of the GTPases Rho and Rac to control convergent extension movements. We find that TRPM7 functions synergistically with non-canonical Wnt signaling to regulate Rac activity. The phenotype caused by depletion of the Ca(2+)- and Mg(2+)-permeant TRPM7 is suppressed by expression of a dominant negative form of Rac, as well as by Mg(2+) supplementation or by expression of the Mg(2+) transporter SLC41A2. Together, these studies demonstrate an essential role for the ion channel TRPM7 and Mg(2+) in Rac-dependent polarized cell movements during vertebrate gastrulation.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures




References
-
- Almonte RA, Heath DL, Whitehall J, Russell MJ, Patole S, Vink R. Gestational magnesium deficiency is deleterious to fetal outcome. Biol Neonate. 1999;76:26–32. - PubMed
-
- Chubanov V, Schlingmann KP, Waring J, Heinzinger J, Kaske S, Waldegger S, Mederos y Schnitzler M, Gudermann T. Hypomagnesemia with secondary hypocalcemia due to a missense mutation in the putative pore-forming region of TRPM6. J Biol Chem. 2007;282:7656–67. - PubMed
-
- Clapham DE. TRP channels as cellular sensors. Nature. 2003;426:517–24. - PubMed
-
- Clark K, Middelbeek J, Dorovkov MV, Figdor CG, Ryazanov AG, Lasonder E, van Leeuwen FN. The alpha-kinases TRPM6 and TRPM7, but not eEF-2 kinase, phosphorylate the assembly domain of myosin IIA, IIB and IIC. FEBS Lett. 2008a;582:2993–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

