Conditional deletion of Atoh1 using Pax2-Cre results in viable mice without differentiated cochlear hair cells that have lost most of the organ of Corti
- PMID: 21146598
- PMCID: PMC3065550
- DOI: 10.1016/j.heares.2010.12.002
Conditional deletion of Atoh1 using Pax2-Cre results in viable mice without differentiated cochlear hair cells that have lost most of the organ of Corti
Abstract
Atonal homolog1 (Atoh1, formerly Math1) is a crucial bHLH transcription factor for inner ear hair cell differentiation. Its absence in embryos results in complete absence of mature hair cells at birth and its misexpression can generate extra hair cells. Thus Atoh1 may be both necessary and sufficient for hair cell differentiation in the ear. Atoh1 null mice die at birth and have some undifferentiated cells in sensory epithelia carrying Atoh1 markers. The fate of these undifferentiated cells in neonates is unknown due to lethality. We use Tg(Pax2-Cre) to delete floxed Atoh1 in the inner ear. This generates viable conditional knockout (CKO) mice for studying the postnatal development of the inner ear without differentiated hair cells. Using in situ hybridization we find that Tg(Pax2-Cre) recombines the floxed Atoh1 prior to detectable Atoh1 expression. Only the posterior canal crista has Atoh1 expressing hair cells due to incomplete recombination. Most of the organ of Corti cells are lost in CKO mice via late embryonic cell death. Marker genes indicate that the organ of Corti is reduced to two rows of cells wedged between flanking markers of the organ of Corti (Fgf10 and Bmp4). These two rows of cells (instead of five rows of supporting cells) are positive for Prox1 in neonates. By postnatal day 14 (P14), the remaining cells of the organ of Corti are transformed into a flat epithelium with no distinction of any specific cell type. However, some of the remaining organ of Corti cells express Myo7a at late postnatal stages and are innervated by remaining afferent fibers. Initial growth of afferents and efferents in embryos shows no difference between control mice and Tg(Pax2-Cre)::Atoh1 CKO mice. Most afferents and efferents are lost in the CKO mutant before birth, except for the apex and few fibers in the base. Afferents focus their projections on patches that express the prosensory specifying gene, Sox2. This pattern of innervation by sensory neurons is maintained at least until P14, but fibers target the few Myo7a positive cells found in later stages.
Copyright © 2010 Elsevier B.V. All rights reserved.
Figures

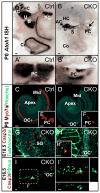
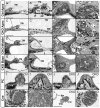
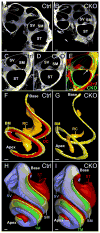




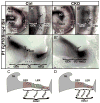
References
-
- Anniko M, Arnesen AR. Cochlear nerve topography and fiber spectrum in the pigmented mouse. Arch Otorhinolaryngol. 1988;245:155–9. - PubMed
-
- Bermingham NA, Hassan BA, Wang VY, Fernandez M, Banfi S, Bellen HJ, Fritzsch B, Zoghbi HY. Proprioceptor pathway development is dependent on Math1. Neuron. 2001;30:411–22. - PubMed
-
- Bermingham NA, Hassan BA, Price SD, Vollrath MA, Ben-Arie N, Eatock RA, Bellen HJ, Lysakowski A, Zoghbi HY. Math1: an essential gene for the generation of inner ear hair cells. Science. 1999;284:1837–41. - PubMed
-
- Campbell JP, Henson MM. Olivocochlear neurons in the brainstem of the mouse. Hear Res. 1988;35:271–4. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

