Negative regulation of ciliary length by ciliary male germ cell-associated kinase (Mak) is required for retinal photoreceptor survival
- PMID: 21148103
- PMCID: PMC3012466
- DOI: 10.1073/pnas.1009437108
Negative regulation of ciliary length by ciliary male germ cell-associated kinase (Mak) is required for retinal photoreceptor survival
Abstract
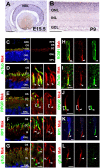
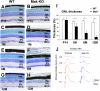
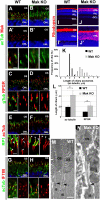
Cilia function as cell sensors in many organs, and their disorders are referred to as "ciliopathies." Although ciliary components and transport machinery have been well studied, regulatory mechanisms of ciliary formation and maintenance are poorly understood. Here we show that male germ cell-associated kinase (Mak) regulates retinal photoreceptor ciliary length and subcompartmentalization. Mak was localized both in the connecting cilia and outer-segment axonemes of photoreceptor cells. In the Mak-null retina, photoreceptors exhibit elongated cilia and progressive degeneration. We observed accumulation of intraflagellar transport 88 (IFT88) and IFT57, expansion of kinesin family member 3A (Kif3a), and acetylated α-tubulin signals in the Mak-null photoreceptor cilia. We found abnormal rhodopsin accumulation in the Mak-null photoreceptor cell bodies at postnatal day 14. In addition, overexpression of retinitis pigmentosa 1 (RP1), a microtubule-associated protein localized in outer-segment axonemes, induced ciliary elongation, and Mak coexpression rescued excessive ciliary elongation by RP1. The RP1 N-terminal portion induces ciliary elongation and increased intensity of acetylated α-tubulin labeling in the cells and is phosphorylated by Mak. These results suggest that Mak is essential for the regulation of ciliary length and is required for the long-term survival of photoreceptors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Ccrk-Mak/Ick signaling is a ciliary transport regulator essential for retinal photoreceptor survival.Life Sci Alliance. 2024 Sep 18;7(11):e202402880. doi: 10.26508/lsa.202402880. Print 2024 Nov. Life Sci Alliance. 2024. PMID: 39293864 Free PMC article.
-
Male germ cell-associated kinase is required for axoneme formation during ciliogenesis in zebrafish photoreceptors.Dis Model Mech. 2024 Jul 1;17(7):dmm050618. doi: 10.1242/dmm.050618. Epub 2024 Jul 16. Dis Model Mech. 2024. PMID: 38813692 Free PMC article.
-
Retinitis pigmentosa caused by mutations in the ciliary MAK gene is relatively mild and is not associated with apparent extra-ocular features.Acta Ophthalmol. 2015 Feb;93(1):83-94. doi: 10.1111/aos.12500. Epub 2014 Nov 11. Acta Ophthalmol. 2015. PMID: 25385675
-
Protein networks and complexes in photoreceptor cilia.Subcell Biochem. 2007;43:209-35. doi: 10.1007/978-1-4020-5943-8_10. Subcell Biochem. 2007. PMID: 17953396 Review.
-
On the Wrong Track: Alterations of Ciliary Transport in Inherited Retinal Dystrophies.Front Cell Dev Biol. 2021 Mar 5;9:623734. doi: 10.3389/fcell.2021.623734. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33748110 Free PMC article. Review.
Cited by
-
CCRK depletion inhibits glioblastoma cell proliferation in a cilium-dependent manner.EMBO Rep. 2013 Aug;14(8):741-7. doi: 10.1038/embor.2013.80. Epub 2013 Jun 7. EMBO Rep. 2013. PMID: 23743448 Free PMC article.
-
ARL13B, a Joubert Syndrome-Associated Protein, Is Critical for Retinogenesis and Elaboration of Mouse Photoreceptor Outer Segments.J Neurosci. 2019 Feb 20;39(8):1347-1364. doi: 10.1523/JNEUROSCI.1761-18.2018. Epub 2018 Dec 20. J Neurosci. 2019. PMID: 30573647 Free PMC article.
-
The Scaffold Protein KATNIP Enhances CILK1 Control of Primary Cilia.Mol Cell Biol. 2023;43(9):472-480. doi: 10.1080/10985549.2023.2246870. Epub 2023 Sep 4. Mol Cell Biol. 2023. PMID: 37665596 Free PMC article.
-
Regulation of cilium length and intraflagellar transport by the RCK-kinases ICK and MOK in renal epithelial cells.PLoS One. 2014 Sep 22;9(9):e108470. doi: 10.1371/journal.pone.0108470. eCollection 2014. PLoS One. 2014. PMID: 25243405 Free PMC article.
-
A murine RP1 missense mutation causes protein mislocalization and slowly progressive photoreceptor degeneration.Am J Pathol. 2014 Oct;184(10):2721-9. doi: 10.1016/j.ajpath.2014.06.010. Epub 2014 Aug 1. Am J Pathol. 2014. PMID: 25088982 Free PMC article.
References
-
- Nigg EA, Raff JW. Centrioles, centrosomes, and cilia in health and disease. Cell. 2009;139:663–678. - PubMed
-
- Fliegauf M, Benzing T, Omran H. When cilia go bad: Cilia defects and ciliopathies. Nat Rev Mol Cell Biol. 2007;8:880–893. - PubMed
-
- Rosenbaum JL, Witman GB. Intraflagellar transport. Nat Rev Mol Cell Biol. 2002;3:813–825. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

