Prescribed dietary phosphate restriction and survival among hemodialysis patients
- PMID: 21148246
- PMCID: PMC3082422
- DOI: 10.2215/CJN.04620510
Prescribed dietary phosphate restriction and survival among hemodialysis patients
Abstract
Background and objectives: Hyperphosphatemia is common among hemodialysis patients. Although prescribed dietary phosphate restriction is a recommended therapy, little is known about the long-term effects on survival.
Design, setting, participants, & measurements: We conducted a post hoc analysis of data from the Hemodialysis Study (n = 1751). Prescribed dietary phosphate was recorded at baseline and annually thereafter. Marginal structural proportional hazard models were fit to estimate the adjusted association between dietary phosphate restriction and mortality in the setting of time-dependent confounding.
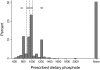
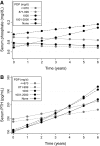
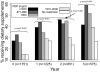
Results: At baseline, prescribed daily phosphate was restricted to levels ≤ 870, 871 to 999, 1000, 1001 to 2000 mg, and not restricted in 300, 314, 307, 297, and 533 participants, respectively. More restrictive prescribed dietary phosphate was associated with poorer indices of nutritional status on baseline analyses and a persistently greater need for nutritional supplementation but not longitudinal changes in caloric or protein intake. On marginal structural analysis, there was a stepwise trend toward greater survival with more liberal phosphate prescription, which reached statistical significance among subjects prescribed 1001 to 2000 mg/d and those with no specified phosphate restriction: hazard ratios (95% CIs) 0.73 (0.54 to 0.97) and 0.71 (0.55 to 0.92), respectively. Subgroup analysis suggested a more pronounced survival benefit of liberal dietary phosphate prescription among nonblacks, participants without hyperphosphatemia, and those not receiving activated vitamin D.
Conclusions: Prescribed dietary phosphate restriction is not associated with improved survival among prevalent hemodialysis patients, and increased level of restriction may be associated with greater mortality particularly in some subgroups.
Figures


Similar articles
-
Phosphate binder use and mortality among hemodialysis patients in the Dialysis Outcomes and Practice Patterns Study (DOPPS): evaluation of possible confounding by nutritional status.Am J Kidney Dis. 2012 Jul;60(1):90-101. doi: 10.1053/j.ajkd.2011.12.025. Epub 2012 Mar 3. Am J Kidney Dis. 2012. PMID: 22385781 Free PMC article.
-
Increased dietary sodium is independently associated with greater mortality among prevalent hemodialysis patients.Kidney Int. 2012 Jul;82(2):204-11. doi: 10.1038/ki.2012.42. Epub 2012 Mar 14. Kidney Int. 2012. PMID: 22418981 Free PMC article. Clinical Trial.
-
Dietary intervention focused on phosphate intake in hemodialysis patients with hyperphosphoremia.Clin Nephrol. 2012 Jun;77(6):476-83. Clin Nephrol. 2012. PMID: 22595390 Clinical Trial.
-
Effect of Phosphate-Specific Diet Therapy on Phosphate Levels in Adults Undergoing Maintenance Hemodialysis: A Systematic Review and Meta-Analysis.Clin J Am Soc Nephrol. 2020 Dec 31;16(1):107-120. doi: 10.2215/CJN.09360620. Clin J Am Soc Nephrol. 2020. PMID: 33380474 Free PMC article.
-
Sevelamer Versus Calcium-Based Binders for Treatment of Hyperphosphatemia in CKD: A Meta-Analysis of Randomized Controlled Trials.Clin J Am Soc Nephrol. 2016 Feb 5;11(2):232-44. doi: 10.2215/CJN.06800615. Epub 2015 Dec 14. Clin J Am Soc Nephrol. 2016. PMID: 26668024 Free PMC article. Review.
Cited by
-
Hyperkalemia in patients undergoing hemodialysis: Its pathophysiology and management.Ther Apher Dial. 2022 Feb;26(1):3-14. doi: 10.1111/1744-9987.13721. Epub 2021 Aug 31. Ther Apher Dial. 2022. PMID: 34378859 Free PMC article. Review.
-
Management of hyperphosphataemia in chronic kidney disease-challenges and solutions.Clin Kidney J. 2013 Apr;6(2):128-36. doi: 10.1093/ckj/sfs173. Epub 2013 Jan 16. Clin Kidney J. 2013. PMID: 26019840 Free PMC article. Review.
-
Hyperphosphatemia in patients with ESRD: assessing the current evidence linking outcomes with treatment adherence.BMC Nephrol. 2013 Jul 18;14:153. doi: 10.1186/1471-2369-14-153. BMC Nephrol. 2013. PMID: 23865421 Free PMC article. Review.
-
Phosphate binder use and mortality among hemodialysis patients in the Dialysis Outcomes and Practice Patterns Study (DOPPS): evaluation of possible confounding by nutritional status.Am J Kidney Dis. 2012 Jul;60(1):90-101. doi: 10.1053/j.ajkd.2011.12.025. Epub 2012 Mar 3. Am J Kidney Dis. 2012. PMID: 22385781 Free PMC article.
-
One-Year Historical Cohort Study of the Phosphate Binder Sucroferric Oxyhydroxide in Patients on Maintenance Hemodialysis.J Ren Nutr. 2019 Sep;29(5):428-437. doi: 10.1053/j.jrn.2018.11.002. Epub 2019 Jan 21. J Ren Nutr. 2019. PMID: 30679076 Free PMC article.
References
-
- Tentori F, Blayney MJ, Albert JM, Gillespie BW, Kerr PG, Bommer J, Young EW, Akizawa T, Akiba T, Pisoni RL, Robinson BM, Port FK: Mortality risk for dialysis patients with different levels of serum calcium, phosphorus, and PTH: The dialysis outcomes and practice patterns study (DOPPS). Am J Kidney Dis 52: 519–530, 2008 - PubMed
-
- Kimata N, Albert JM, Akiba T, Yamazaki S, Kawaguchi T, Fukuhara S, Akizawa T, Saito A, Asano Y, Kurokawa K, Pisoni RL, Port FK: Association of mineral metabolism factors with all-cause and cardiovascular mortality in hemodialysis patients: The Japan dialysis outcomes and practice patterns study. Hemodial Int 11: 340–348, 2007 - PubMed
-
- Young EW, Albert JM, Satayathum S, Goodkin DA, Pisoni RL, Akiba T, Akizawa T, Kurokawa K, Bommer J, Piera L, Port FK: Predictors and consequences of altered mineral metabolism: The dialysis outcomes and practice patterns study. Kidney Int 67: 1179–1187, 2005 - PubMed
-
- Slinin Y, Foley RN, Collins AJ: Calcium, phosphorus, parathyroid hormone, and cardiovascular disease in hemodialysis patients: The USRDS waves 1, 3, and 4 study. J Am Soc Nephrol 16: 1788–1793, 2005 - PubMed
-
- Port FK, Pisoni RL, Bommer J, Locatelli F, Jadoul M, Eknoyan G, Kurokawa K, Canaud BJ, Finley MP, Young EW: Improving outcomes for dialysis patients in the international dialysis outcomes and practice patterns study. Clin J Am Soc Nephrol 1: 246–255, 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

