Structural and biochemical elucidation of mechanism for decarboxylative condensation of beta-keto acid by curcumin synthase
- PMID: 21148316
- PMCID: PMC3057783
- DOI: 10.1074/jbc.M110.196279
Structural and biochemical elucidation of mechanism for decarboxylative condensation of beta-keto acid by curcumin synthase
Abstract
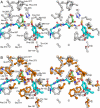
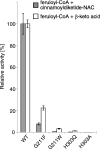
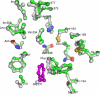
The typical reaction catalyzed by type III polyketide synthases (PKSs) is a decarboxylative condensation between acyl-CoA (starter substrate) and malonyl-CoA (extender substrate). In contrast, curcumin synthase 1 (CURS1), which catalyzes curcumin synthesis by condensing feruloyl-CoA with a diketide-CoA, uses a β-keto acid (which is derived from diketide-CoA) as an extender substrate. Here, we determined the crystal structure of CURS1 at 2.32 Å resolution. The overall structure of CURS1 was very similar to the reported structures of type III PKSs and exhibited the αβαβα fold. However, CURS1 had a unique hydrophobic cavity in the CoA-binding tunnel. Replacement of Gly-211 with Phe greatly reduced the enzyme activity. The crystal structure of the G211F mutant (at 2.5 Å resolution) revealed that the side chain of Phe-211 occupied the hydrophobic cavity. Biochemical studies demonstrated that CURS1 catalyzes the decarboxylative condensation of a β-keto acid using a mechanism identical to that for normal decarboxylative condensation of malonyl-CoA by typical type III PKSs. Furthermore, the extender substrate specificity of CURS1 suggested that hydrophobic interaction between CURS1 and a β-keto acid may be important for CURS1 to use an extender substrate lacking the CoA moiety. From these results and a modeling study on substrate binding, we concluded that the hydrophobic cavity is responsible for the hydrophobic interaction between CURS1 and a β-keto acid, and this hydrophobic interaction enables the β-keto acid moiety to access the catalytic center of CURS1 efficiently.
Figures


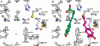

References
-
- Austin M. B., Noel J. P. (2003) Nat. Prod. Rep. 20, 79–110 - PubMed
-
- Abe I., Morita H. (2010) Nat. Prod. Rep. 27, 809–838 - PubMed
-
- Ferrer J. L., Jez J. M., Bowman M. E., Dixon R. A., Noel J. P. (1999) Nat. Struct. Biol. 6, 775–784 - PubMed
-
- Jez J. M., Austin M. B., Ferrer J., Bowman M. E., Schröder J., Noel J. P. (2000) Chem. Biol. 7, 919–930 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

