RNAi screen identifies a role for adaptor protein AP-3 in sorting to the regulated secretory pathway
- PMID: 21149569
- PMCID: PMC3002028
- DOI: 10.1083/jcb.201006131
RNAi screen identifies a role for adaptor protein AP-3 in sorting to the regulated secretory pathway
Abstract
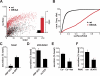
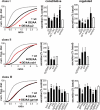
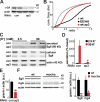
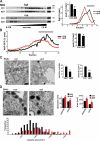
The regulated release of proteins depends on their inclusion within large dense-core vesicles (LDCVs) capable of regulated exocytosis. LDCVs form at the trans-Golgi network (TGN), but the mechanism for protein sorting to this regulated secretory pathway (RSP) and the cytosolic machinery involved in this process have remained poorly understood. Using an RNA interference screen in Drosophila melanogaster S2 cells, we now identify a small number of genes, including several subunits of the heterotetrameric adaptor protein AP-3, which are required for sorting to the RSP. In mammalian neuroendocrine cells, loss of AP-3 dysregulates exocytosis due to a primary defect in LDCV formation. Previous work implicated AP-3 in the endocytic pathway, but we find that AP-3 promotes sorting to the RSP within the biosynthetic pathway at the level of the TGN. Although vesicles with a dense core still form in the absence of AP-3, they contain substantially less synaptotagmin 1, indicating that AP-3 concentrates the proteins required for regulated exocytosis.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

