IL-6-dependent PGE2 secretion by mesenchymal stem cells inhibits local inflammation in experimental arthritis
- PMID: 21151872
- PMCID: PMC2998425
- DOI: 10.1371/journal.pone.0014247
IL-6-dependent PGE2 secretion by mesenchymal stem cells inhibits local inflammation in experimental arthritis
Abstract
Background: Based on their capacity to suppress immune responses, multipotent mesenchymal stromal cells (MSC) are intensively studied for various clinical applications. Although it has been shown in vitro that the immunomodulatory effect of MSCs mainly occurs through the secretion of soluble mediators, the mechanism is still not completely understood. The aim of the present study was to better understand the mechanisms underlying the suppressive effect of MSCs in vivo, using cells isolated from mice deficient in the production of inducible nitric oxide synthase (iNOS) or interleukin (IL)-6 in the murine model of collagen-induced arthritis.
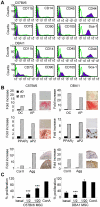
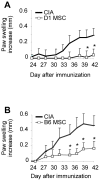
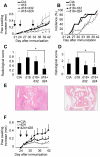
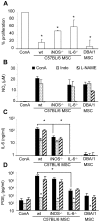
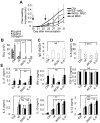
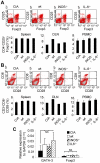
Principal findings: In the present study, we show that primary murine MSCs from various strains of mice or isolated from mice deficient for iNOS or IL-6 exhibit different immunosuppressive potential. The immunomodulatory function of MSCs was mainly attributed to IL-6-dependent secretion of prostaglandin E2 (PGE2) with a minor role for NO. To address the role of these molecules in vivo, we used the collagen-induced arthritis as an experimental model of immune-mediated disorder. MSCs effectively inhibited collagen-induced inflammation during a narrow therapeutic window. In contrast to wild type MSCs, IL-6-deficient MSCs and to a lesser extent iNOS-deficient MSCs were not able to reduce the clinical signs of arthritis. Finally, we show that, independently of NO or IL-6 secretion or Treg cell induction, MSCs modulate the host response by inducing a switch to a Th2 immune response.
Significance: Our data indicate that mscs mediate their immunosuppressive effect via two modes of action: locally, they reduce inflammation through the secretion of anti-proliferative mediators, such as NO and mainly PGE2, and systemically they switch the host response from a Th1/Th17 towards a Th2 immune profile.
Conflict of interest statement
Figures
References
-
- Vinatier C, Mrugala D, Jorgensen C, Guicheux J, Noel D. Cartilage engineering: a crucial combination of cells, biomaterials and biofactors. Trends Biotechnol. 2009;27:307–314. - PubMed
-
- Djouad F, Bouffi C, Ghannam S, Noel D, Jorgensen C. Mesenchymal stem cells: innovative therapeutic tools for rheumatic diseases. Nat Rev Rheumatol. 2009;5:392–399. - PubMed
-
- Spaggiari GM, Capobianco A, Abdelrazik H, Becchetti F, Mingari MC, et al. Mesenchymal stem cells inhibit natural killer-cell proliferation, cytotoxicity, and cytokine production: role of indoleamine 2,3-dioxygenase and prostaglandin E2. Blood. 2008;111:1327–1333. - PubMed
-
- Sato K, Ozaki K, Oh I, Meguro A, Hatanaka K, et al. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood. 2007;109:228–234. - PubMed
-
- Ren G, Zhang L, Zhao X, Xu G, Zhang Y, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2:141–150. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

