Fusion between Leishmania amazonensis and Leishmania major parasitophorous vacuoles: live imaging of coinfected macrophages
- PMID: 21151877
- PMCID: PMC2998430
- DOI: 10.1371/journal.pntd.0000905
Fusion between Leishmania amazonensis and Leishmania major parasitophorous vacuoles: live imaging of coinfected macrophages
Abstract
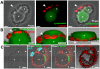
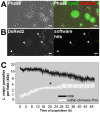
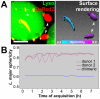
Protozoan parasites of the genus Leishmania alternate between flagellated, elongated extracellular promastigotes found in insect vectors, and round-shaped amastigotes enclosed in phagolysosome-like Parasitophorous Vacuoles (PVs) of infected mammalian host cells. Leishmania amazonensis amastigotes occupy large PVs which may contain many parasites; in contrast, single amastigotes of Leishmania major lodge in small, tight PVs, which undergo fission as parasites divide. To determine if PVs of these Leishmania species can fuse with each other, mouse macrophages in culture were infected with non-fluorescent L. amazonensis amastigotes and, 48 h later, superinfected with fluorescent L. major amastigotes or promastigotes. Fusion was investigated by time-lapse image acquisition of living cells and inferred from the colocalization of parasites of the two species in the same PVs. Survival, multiplication and differentiation of parasites that did or did not share the same vacuoles were also investigated. Fusion of PVs containing L. amazonensis and L. major amastigotes was not found. However, PVs containing L. major promastigotes did fuse with pre-established L. amazonensis PVs. In these chimeric vacuoles, L. major promastigotes remained motile and multiplied, but did not differentiate into amastigotes. In contrast, in doubly infected cells, within their own, unfused PVs metacyclic-enriched L. major promastigotes, but not log phase promastigotes--which were destroyed--differentiated into proliferating amastigotes. The results indicate that PVs, presumably customized by L. major amastigotes or promastigotes, differ in their ability to fuse with L. amazonensis PVs. Additionally, a species-specific PV was required for L. major destruction or differentiation--a requirement for which mechanisms remain unknown. The observations reported in this paper should be useful in further studies of the interactions between PVs to different species of Leishmania parasites, and of the mechanisms involved in the recognition and fusion of PVs.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Rabinovitch M, Veras PS. Cohabitation of Leishmania amazonensis and Coxiella burnetii. Trends Microbiol. 1996;4:158–161. - PubMed
-
- de Chastellier C, Thibon M, Rabinovitch M. Construction of chimeric phagosomes that shelter Mycobacterium avium and Coxiella burnetii (phase II) in doubly infected mouse macrophages: an ultrastructural study. Eur J Cell Biol. 1999;78:580–592. - PubMed
-
- Andreoli WK, Taniwaki NN, Mortara RA. Survival of Trypanosoma cruzi metacyclic trypomastigotes within Coxiella burnetii vacuoles: differentiation and replication within an acidic milieu. Microbes Infect. 2006;8:172–182. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

