Long-range regulatory synergy is required to allow control of the TAC1 locus by MEK/ERK signalling in sensory neurones
- PMID: 21160161
- PMCID: PMC3718575
- DOI: 10.1159/000322010
Long-range regulatory synergy is required to allow control of the TAC1 locus by MEK/ERK signalling in sensory neurones
Abstract
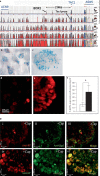
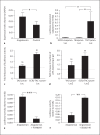
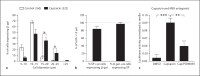
Changes in the expression of the neuropeptide substance P (SP) in different populations of sensory neurones are associated with the progression of chronic inflammatory disease. Thus, understanding the genomic and cellular mechanisms driving the expression of the TAC1 gene, which encodes SP, in sensory neurones is essential to understanding its role in inflammatory disease. We used a novel combination of computational genomics, primary-cell culture and mouse transgenics to determine the genomic and cellular mechanisms that control the expression of TAC1 in sensory neurones. Intriguingly, we demonstrated that the promoter of the TAC1 gene must act in synergy with a remote enhancer, identified using comparative genomics, to respond to MAPK signalling that modulates the expression of TAC1 in sensory neurones. We also reveal that noxious stimulation of sensory neurones triggers this synergy in larger diameter sensory neurones--an expression of SP associated with hyperalgesia. This noxious stimulation of TAC1 enhancer-promotor synergy could be strongly blocked by antagonism of the MEK pathway. This study provides a unique insight into the role of long-range enhancer-promoter synergy and selectivity in the tissue-specific response of promoters to specific signal transduction pathways and suggests a possible new avenue for the development of novel anti-inflammatory therapies.
Copyright © 2010 S. Karger AG, Basel.
Figures


References
-
- Geppetti P, Nassini R, Materazzi S, Benemei S. The concept of neurogenic inflammation. BJU Int. 2008;101(Suppl 3):2–6. - PubMed
-
- Cao YQ, Mantyh PW, Carlson EJ, Gillespie AM, Epstein CJ, Basbaum AI. Primary afferent tachykinins are required to experience moderate to intense pain. Nature. 1998;392:390–394. - PubMed
-
- Hokfelt T, Pernow B, Wahren J. Substance p: a pioneer amongst neuropeptides. J Intern Med. 2001;249:27–40. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

