Alkaloids from stems of Esenbeckia leiocarpa Engl. (Rutaceae) as potential treatment for Alzheimer disease
- PMID: 21160449
- PMCID: PMC6259197
- DOI: 10.3390/molecules15129205
Alkaloids from stems of Esenbeckia leiocarpa Engl. (Rutaceae) as potential treatment for Alzheimer disease
Abstract
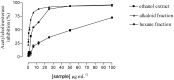
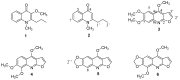
Esenbeckia leiocarpa Engl. (Rutaceae), popularly known as guarantã, goiabeira, is a native tree from Brazil. Bioactivity-guided fractionation of the ethanol stems extract afforded the isolation of six alkaloids: leiokinine A, leptomerine, kokusaginine, skimmianine, maculine and flindersiamine. All isolated compounds were tested for acetyl cholinesterase inhibition, in vitro and displayed anticholinesterasic activity. The alkaloid leptomerine showed the highest activity (IC₅₀ = 2.5 mM), similar to that of the reference compound galanthamine (IC₅₀ = 1.7 mM). The results showed for the first time the presence of alkaloids leptomerine and skimmianine in E. leiocarpa (Engl.) with potent anticholinesterasic activity.
Figures

References
-
- Barros-Filho B.A., Nunes F.M., Oliveira M.C.F., Andrade-Neto M., Mattos M.C., Barbosa F.G., Mafezoli J., Pirani J.R. Secondary Metabolites of Esenbeckia almawillia Kaastra (Rutaceae) Quím. Nova. 2007;30:1589–1591. doi: 10.1590/S0100-40422007000700017. - DOI
-
- Guilhon G.M.S.P., Baetas A.C.S., Maia J.G.S., Conserva L.M. 2-alkyl-4-quinolone alkaloids and cinnamic acid derivatives from Esenbeckia almawillia. Phytochemistry. 1994;37:1193.
-
- Johnstone R.A.W., Rose M.E. A rapid, simple and mild procedure for alkylation of phenols, alcohols, amides and acids. Tetrahedron. 1979;35:2169–2173. doi: 10.1016/0040-4020(79)87035-0. - DOI
-
- Lorenzi H. Brazilian Trees: A Guide to the Identification and Cultivation of Brazilian Native Trees. Plantarum; Nova Odessa, SP, Brazil: 1992. p. 307.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

