Prostaglandin dehydrogenase (PGDH) in granulosa cells of primate periovulatory follicles is regulated by the ovulatory gonadotropin surge via multiple G proteins
- PMID: 21167905
- PMCID: PMC3039104
- DOI: 10.1016/j.mce.2010.12.016
Prostaglandin dehydrogenase (PGDH) in granulosa cells of primate periovulatory follicles is regulated by the ovulatory gonadotropin surge via multiple G proteins
Abstract
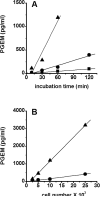
The ovulatory gonadotropin surge increases granulosa cell prostaglandin synthesis as well as prostaglandin dehydrogenase (PGDH), the key enzyme responsible for prostaglandin metabolism. To investigate gonadotropin regulation of PGDH in the primate follicle, monkey granulosa cells were obtained across the 40-h periovulatory interval. PGDH activity was low before the ovulatory hCG stimulus, peaked 12-24 h after hCG, and was low again 36 h after hCG administration. Granulosa cells maintained in vitro with hCG showed a similar temporal pattern of PGDH. The LH/CG receptor can utilize multiple signaling pathways to regulate intracellular events. Gonadotropin-stimulated cAMP appears to act primarily via the Epacs to increase PGDH mRNA, protein, and activity. In contrast, PLC activation of PKC likely decreases PGDH mRNA, protein, and activity late in the periovulatory interval. Increased, then decreased PGDH activity may delay accumulation of prostaglandins in the follicle until late in the periovulatory interval, contributing to timely ovulation in primates.
Copyright © 2010 Elsevier Ireland Ltd. All rights reserved.
Figures

Similar articles
-
Cloning of equine prostaglandin dehydrogenase and its gonadotropin-dependent regulation in theca and mural granulosa cells of equine preovulatory follicles during the ovulatory process.Reproduction. 2007 Feb;133(2):455-66. doi: 10.1530/REP-06-0210. Reproduction. 2007. PMID: 17307913
-
Prostaglandin dehydrogenase and prostaglandin levels in periovulatory follicles: implications for control of primate ovulation by prostaglandin E2.J Clin Endocrinol Metab. 2005 Feb;90(2):1021-7. doi: 10.1210/jc.2004-1229. Epub 2004 Nov 2. J Clin Endocrinol Metab. 2005. PMID: 15522934
-
An ovulatory gonadotropin stimulus increases cytosolic phospholipase A2 expression and activity in granulosa cells of primate periovulatory follicles.J Clin Endocrinol Metab. 2005 Oct;90(10):5858-65. doi: 10.1210/jc.2005-0980. Epub 2005 Jun 21. J Clin Endocrinol Metab. 2005. PMID: 15972573
-
Adipose differentiation-related protein: a gonadotropin- and prostaglandin-regulated protein in primate periovulatory follicles.Biol Reprod. 2005 Jun;72(6):1305-14. doi: 10.1095/biolreprod.104.037523. Epub 2005 Feb 2. Biol Reprod. 2005. PMID: 15689536
-
Two pathways for prostaglandin F2 alpha synthesis by the primate periovulatory follicle.Reproduction. 2008 Jul;136(1):53-63. doi: 10.1530/REP-07-0514. Epub 2008 Apr 4. Reproduction. 2008. PMID: 18390687 Free PMC article.
Cited by
-
Novel contraceptive targets to inhibit ovulation: the prostaglandin E2 pathway.Hum Reprod Update. 2015 Sep-Oct;21(5):652-70. doi: 10.1093/humupd/dmv026. Epub 2015 May 29. Hum Reprod Update. 2015. PMID: 26025453 Free PMC article. Review.
-
Ovarian reserve status in young women is associated with altered gene expression in membrana granulosa cells.Mol Hum Reprod. 2012 Jul;18(7):362-71. doi: 10.1093/molehr/gas008. Epub 2012 Feb 20. Mol Hum Reprod. 2012. PMID: 22355044 Free PMC article.
References
-
- Casciani V, Marinoni E, Bocking AD, Moscarini M, Di Iorio R, Challis JR. Opposite effect of phorbol ester PMA on PTGS2 and PGDH mRNA expression in human chorion trophoblast cells. Reproductive Sciences. 2008;15:40–50. - PubMed
-
- Chaffin CL, Hess DL, Stouffer RL. Dynamics of periovulatory steroidogenesis in the rhesus monkey follicle after controlled ovarian stimulation. Human Reproduction. 1999;14:642–649. - PubMed
-
- Chaffin CL, Stouffer RL. Expression of matrix metalloproteinases and their tissue inhibitor messenger ribonucleic acids in macaque periovulatory granulosa cells: Time course and steroid regulation. Biology of Reproduction. 1999;61:14–21. - PubMed
-
- Chin EC, Abayasekara DR. Progersterone secretion by luteinizing human granulosa cells: a possible cAMP-dependent but PKA-independent mechanism involved in its regulation. Journal of Endocrinology. 2004;183:51–60. - PubMed
-
- Cho H, Tai HH. Inhibition of NAD+-dependent 15-hydroxyprostaglandin dehydrogenase (15-PGDH) by cyclooxygenase inhibitors and chemopreventive agents. Prostaglandins, Leukotrienes, and Essential Fatty Acids. 2002;67:461–465. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

