Respiratory modulation of the pharyngeal airway in lean and obese mice
- PMID: 21167963
- PMCID: PMC3032032
- DOI: 10.1016/j.resp.2010.12.006
Respiratory modulation of the pharyngeal airway in lean and obese mice
Abstract
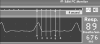
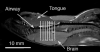
Obesity is an important risk factor for pharyngeal airway collapse in obstructive sleep apnea (OSA). To examine the effect of obesity on pharyngeal airway size on inspiration and expiration, respiratory-gated MRI of the pharynx was compared in New Zealand obese (NZO) and New Zealand white (NZW) mice (weights: 50.4g vs. 34.7g, p<0.0001).
Results: (1) pharyngeal airway cross-sectional area was greater during inspiration than expiration in NZO mice, but in NZW mice airway area was greater in expiration than inspiration; (2) inspiratory-to-expiratory changes in both mouse strains were largest in the caudal pharynx; and (3) during expiration, airway size tended to be larger, though non-significantly, in NZW than NZO mice. The respiratory pattern differences are likely attributable to obesity that is the main difference between NZO and NZW mice. The data support an hypothesis that pharyngeal airway patency in obesity is dependent on inspiratory dilation and may be vulnerable to loss of neuromuscular pharyngeal activation.
Copyright © 2011 Elsevier B.V. All rights reserved.
Figures





Similar articles
-
Altered upper airway and soft tissue structures in the New Zealand Obese mouse.Am J Respir Crit Care Med. 2009 Jan 15;179(2):158-69. doi: 10.1164/rccm.200809-1435OC. Epub 2008 Nov 7. Am J Respir Crit Care Med. 2009. PMID: 18996996 Free PMC article.
-
Phasic respiratory pharyngeal mechanics by magnetic resonance imaging in lean and obese zucker rats.Am J Respir Crit Care Med. 2006 May 1;173(9):1031-7. doi: 10.1164/rccm.200505-705OC. Epub 2006 Jan 26. Am J Respir Crit Care Med. 2006. PMID: 16439721 Free PMC article.
-
Changes in pharyngeal collapsibility and genioglossus reflex responses to negative pressure during the respiratory cycle in obstructive sleep apnoea.J Physiol. 2020 Feb;598(3):567-580. doi: 10.1113/JP278433. Epub 2020 Jan 15. J Physiol. 2020. PMID: 31782971
-
Obesity and obstructive sleep apnoea: mechanisms for increased collapsibility of the passive pharyngeal airway.Respirology. 2012 Jan;17(1):32-42. doi: 10.1111/j.1440-1843.2011.02093.x. Respirology. 2012. PMID: 22023094 Review.
-
Obesity and upper airway control during sleep.J Appl Physiol (1985). 2010 Feb;108(2):430-5. doi: 10.1152/japplphysiol.00919.2009. Epub 2009 Oct 29. J Appl Physiol (1985). 2010. PMID: 19875707 Free PMC article. Review.
Cited by
-
A Novel Non-invasive Approach for Measuring Upper Airway Collapsibility in Mice.Front Neurol. 2018 Nov 20;9:985. doi: 10.3389/fneur.2018.00985. eCollection 2018. Front Neurol. 2018. PMID: 30524362 Free PMC article.
-
Novel whole body plethysmography system for the continuous characterization of sleep and breathing in a mouse.J Appl Physiol (1985). 2012 Feb;112(4):671-80. doi: 10.1152/japplphysiol.00818.2011. Epub 2011 Dec 1. J Appl Physiol (1985). 2012. PMID: 22134700 Free PMC article.
-
Cardiovascular consequences of sleep apnea.Lung. 2012 Apr;190(2):113-32. doi: 10.1007/s00408-011-9340-1. Epub 2011 Nov 3. Lung. 2012. PMID: 22048845 Review.
-
Sleep-disordered breathing in C57BL/6J mice with diet-induced obesity.Sleep. 2018 Aug 1;41(8):zsy089. doi: 10.1093/sleep/zsy089. Sleep. 2018. PMID: 29767787 Free PMC article.
-
Animal models of sleep disorders.Comp Med. 2013 Apr;63(2):91-104. Comp Med. 2013. PMID: 23582416 Free PMC article. Review.
References
-
- Bartlett D., Jr. Upper airway motor systems. In: Fishman AP, editor. The Respiratory System. 2nd edn American Physiology Society; Baltimore: 1986. pp. 223–246.
-
- Bielschowsky M, Goodall CM. Origin of inbred NZ mouse strains. Cancer Res. 1970;30:834–836. - PubMed
-
- Bonora M, Bartlett D, Jr., Knuth SL. Changes in upper airway muscle activity related to head position in awake cats. Respir Physiol. 1985;60:181–192. - PubMed
-
- Brennick MJ, Pack AI, Pickup S, Schwab RJ. Cine-MRI shows airway inspiratory and expiratory compromise in New Zealand obese mice. Am J Respir Crit Care Med. 2007;175:A754.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

