Complement receptor Mac-1 is an adaptor for NB1 (CD177)-mediated PR3-ANCA neutrophil activation
- PMID: 21193407
- PMCID: PMC3044964
- DOI: 10.1074/jbc.M110.171256
Complement receptor Mac-1 is an adaptor for NB1 (CD177)-mediated PR3-ANCA neutrophil activation
Abstract
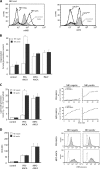
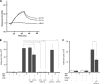
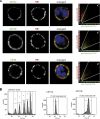
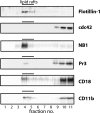
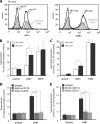
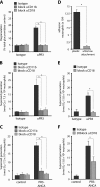
The glycosylphosphatidylinositol (GPI)-anchored neutrophil-specific receptor NB1 (CD177) presents the autoantigen proteinase 3 (PR3) on the membrane of a neutrophil subset. PR3-ANCA-activated neutrophils participate in small-vessel vasculitis. Since NB1 lacks an intracellular domain, we characterized components of the NB1 signaling complex that are pivotal for neutrophil activation. PR3-ANCA resulted in degranulation and superoxide production in the mNB1(pos)/PR3(high) neutrophils, but not in the mNB1(neg)/PR3(low) subset, whereas MPO-ANCA and fMLP caused similar responses. The NB1 signaling complex that was precipitated from plasma membranes contained the transmembrane receptor Mac-1 (CD11b/CD18) as shown by MS/MS analysis and immunoblotting. NB1 co-precipitation was less for CD11a and not detectable for CD11c. NB1 showed direct protein-protein interactions with both CD11b and CD11a by surface plasmon resonance analysis (SPR). However, when these integrins were presented as heterodimeric transmembrane proteins on transfected cells, only CD11b/CD18 (Mac-1)-transfected cells adhered to immobilized NB1 protein. This adhesion was inhibited by mAb against NB1, CD11b, and CD18. NB1, PR3, and Mac-1 were located within lipid rafts. In addition, confocal microscopy showed the strongest NB1 co-localization with CD11b and CD18 on the neutrophil. Stimulation with NB1-activating mAb triggered degranulation and superoxide production in mNB1(pos)/mPR3(high) neutrophils, and this effect was reduced using blocking antibodies to CD11b. CD11b blockade also inhibited PR3-ANCA-induced neutrophil activation, even when β2-integrin ligand-dependent signals were omitted. We establish the pivotal role of the NB1-Mac-1 receptor interaction for PR3-ANCA-mediated neutrophil activation.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

