Phenotypic and genotypic evidence for L-fucose utilization by Campylobacter jejuni
- PMID: 21193610
- PMCID: PMC3067607
- DOI: 10.1128/JB.01252-10
Phenotypic and genotypic evidence for L-fucose utilization by Campylobacter jejuni
Abstract
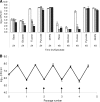
Campylobacter jejuni remains among the leading causes of bacterial food-borne illness. The current understanding of Campylobacter physiology suggests that it is asaccharolytic and is unable to catabolize exogenous carbohydrates. Contrary to this paradigm, we provide evidence for l-fucose utilization by C. jejuni. The fucose phenotype, shown in chemically defined medium, is strain specific and linked to an 11-open reading frame (ORF) plasticity region of the bacterial chromosome. By constructing a mutation in fucP (encoding a putative fucose permease), one of the genes in the plasticity region, we found that this locus is required for fucose utilization. Consistent with their function in fucose utilization, transcription of the genes in the locus is highly inducible by fucose. PCR screening revealed a broad distribution of this genetic locus in strains derived from various host species, and the presence of this locus was consistently associated with fucose utilization. Birds inoculated with the fucP mutant strain alone were colonized at a level comparable to that by the wild-type strain; however, in cocolonization experiments, the mutant was significantly outcompeted by the wild-type strain when birds were inoculated with a low dose (10⁵ CFU per bird). This advantage was not observed when birds were inoculated at a higher inoculum dose (10⁸ CFU per bird). These results demonstrated a previously undescribed substrate that supports growth of C. jejuni and identified the genetic locus associated with the utilization of this substrate. These findings substantially enhance our understanding of the metabolic repertoire of C. jejuni and the role of metabolic diversity in Campylobacter pathobiology.
Figures


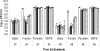


Similar articles
-
L-fucose utilization provides Campylobacter jejuni with a competitive advantage.Proc Natl Acad Sci U S A. 2011 Apr 26;108(17):7194-9. doi: 10.1073/pnas.1014125108. Epub 2011 Apr 11. Proc Natl Acad Sci U S A. 2011. PMID: 21482772 Free PMC article.
-
L-fucose influences chemotaxis and biofilm formation in Campylobacter jejuni.Mol Microbiol. 2016 Aug;101(4):575-89. doi: 10.1111/mmi.13409. Epub 2016 Jun 10. Mol Microbiol. 2016. PMID: 27145048
-
Host associations of Campylobacter jejuni and Campylobacter coli isolates carrying the L-fucose or d-glucose utilization cluster.Int J Food Microbiol. 2024 Dec 2;425:110855. doi: 10.1016/j.ijfoodmicro.2024.110855. Epub 2024 Aug 5. Int J Food Microbiol. 2024. PMID: 39191191
-
Phenotypic and genotypic methods for typing Campylobacter jejuni and Campylobacter coli in poultry.Poult Sci. 2012 Jan;91(1):255-64. doi: 10.3382/ps.2011-01414. Poult Sci. 2012. PMID: 22184452 Review.
-
Re-thinking the chicken-Campylobacter jejuni interaction: a review.Avian Pathol. 2018 Aug;47(4):352-363. doi: 10.1080/03079457.2018.1475724. Epub 2018 Jun 11. Avian Pathol. 2018. PMID: 29764197 Review.
Cited by
-
Activation of the l-fucose utilization cluster in Campylobacter jejuni induces proteomic changes and enhances Caco-2 cell invasion and fibronectin binding.Heliyon. 2024 Jul 26;10(16):e34996. doi: 10.1016/j.heliyon.2024.e34996. eCollection 2024 Aug 30. Heliyon. 2024. PMID: 39220920 Free PMC article.
-
The Role of luxS in Campylobacter jejuni Beyond Intercellular Signaling.Microbiol Spectr. 2023 Feb 1;11(2):e0257222. doi: 10.1128/spectrum.02572-22. Online ahead of print. Microbiol Spectr. 2023. PMID: 36722966 Free PMC article.
-
Genome and Methylome analysis of a phylogenetic novel Campylobacter coli cluster with C. jejuni introgression.Microb Genom. 2021 Oct;7(10):000679. doi: 10.1099/mgen.0.000679. Microb Genom. 2021. PMID: 34661518 Free PMC article.
-
Emergence of fexA in Mediating Resistance to Florfenicols in Campylobacter.Antimicrob Agents Chemother. 2020 Jun 23;64(7):e00260-20. doi: 10.1128/AAC.00260-20. Print 2020 Jun 23. Antimicrob Agents Chemother. 2020. PMID: 32366706 Free PMC article.
-
Enzyme IIANtr Regulates Salmonella Invasion Via 1,2-Propanediol And Propionate Catabolism.Sci Rep. 2017 Mar 23;7:44827. doi: 10.1038/srep44827. Sci Rep. 2017. PMID: 28333132 Free PMC article.
References
-
- Allen, K. J., and M. W. Griffiths. 2001. Effect of environmental and chemotactic stimuli on the activity of the Campylobacter jejuni flaA sigma(28) promoter. FEMS Microbiol. Lett. 205:43-48. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

