Fenamates as TRP channel blockers: mefenamic acid selectively blocks TRPM3
- PMID: 21198543
- PMCID: PMC3081119
- DOI: 10.1111/j.1476-5381.2010.01186.x
Fenamates as TRP channel blockers: mefenamic acid selectively blocks TRPM3
Abstract
Background and purpose: Fenamates are N-phenyl-substituted anthranilic acid derivatives clinically used as non-steroid anti-inflammatory drugs in pain treatment. Reports describing fenamates as tools to interfere with cellular volume regulation attracted our attention based on our interest in the role of the volume-modulated transient receptor potential (TRP) channels TRPM3 and TRPV4.
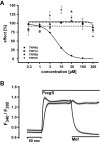
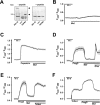
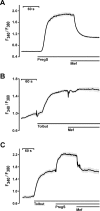
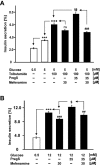
Experimental approach: Firstly, we measured the blocking potencies and selectivities of fenamates on TRPM3 and TRPV4 as well as TRPC6 and TRPM2 by Ca(2+) imaging in the heterologous HEK293 cell system. Secondly, we further investigated the effects of mefenamic acid on cytosolic Ca(2+) and on the membrane voltage in single HEK293 cells that exogenously express TRPM3. Thirdly, in insulin-secreting INS-1E cells, which endogenously express TRPM3, we validated the effect of mefenamic acid on cytosolic Ca(2+) and insulin secretion.
Key results: We identified and characterized mefenamic acid as a selective and potent TRPM3 blocker, whereas other fenamate structures non-selectively blocked TRPM3, TRPV4, TRPC6 and TRPM2.
Conclusions and implications: This study reveals that mefenamic acid selectively inhibits TRPM3-mediated calcium entry. This selectivity was further confirmed using insulin-secreting cells. K(ATP) channel-dependent increases in cytosolic Ca(2+) and insulin secretion were not blocked by mefenamic acid, but the selective stimulation of TRPM3-dependent Ca(2+) entry and insulin secretion induced by pregnenolone sulphate were inhibited. However, the physiological regulator of TRPM3 in insulin-secreting cells remains to be elucidated, as well as the conditions under which the inhibition of TRPM3 can impair pancreatic β-cell function. Our results strongly suggest mefenamic acid is the most selective fenamate to interfere with TRPM3 function.
© 2011 The Authors. British Journal of Pharmacology © 2011 The British Pharmacological Society.
Figures


Similar articles
-
Pharmacological comparison of novel synthetic fenamate analogues with econazole and 2-APB on the inhibition of TRPM2 channels.Br J Pharmacol. 2012 Nov;167(6):1232-43. doi: 10.1111/j.1476-5381.2012.02058.x. Br J Pharmacol. 2012. PMID: 22646516 Free PMC article.
-
Effect of non-steroidal anti-inflammatory drugs and new fenamate analogues on TRPC4 and TRPC5 channels.Biochem Pharmacol. 2012 Apr 1;83(7):923-31. doi: 10.1016/j.bcp.2012.01.014. Epub 2012 Jan 20. Biochem Pharmacol. 2012. PMID: 22285229
-
Activation of TRPA1 channels by fenamate nonsteroidal anti-inflammatory drugs.Pflugers Arch. 2010 Mar;459(4):579-92. doi: 10.1007/s00424-009-0749-9. Epub 2009 Nov 4. Pflugers Arch. 2010. PMID: 19888597 Free PMC article.
-
The potential and promise of mefenamic acid.Expert Rev Clin Pharmacol. 2013 May;6(3):289-305. doi: 10.1586/ecp.13.15. Expert Rev Clin Pharmacol. 2013. PMID: 23656341 Review.
-
Signal transduction via TRPM3 channels in pancreatic β-cells.J Mol Endocrinol. 2013 Apr 23;50(3):R75-83. doi: 10.1530/JME-12-0237. Print 2013 Jun. J Mol Endocrinol. 2013. PMID: 23511953 Review.
Cited by
-
Flufenamic acid as an ion channel modulator.Pharmacol Ther. 2013 May;138(2):272-84. doi: 10.1016/j.pharmthera.2013.01.012. Epub 2013 Jan 25. Pharmacol Ther. 2013. PMID: 23356979 Free PMC article. Review.
-
In silico screening, synthesis, characterization and biological evaluation of novel anticancer agents as potential COX-2 inhibitors.Daru. 2023 Dec;31(2):119-133. doi: 10.1007/s40199-023-00467-x. Epub 2023 Jul 15. Daru. 2023. PMID: 37454036 Free PMC article.
-
Diclofenac, a nonsteroidal anti-inflammatory drug, is an antagonist of human TRPM3 isoforms.Pharmacol Res Perspect. 2016 Apr 7;4(3):e00232. doi: 10.1002/prp2.232. eCollection 2016 Jun. Pharmacol Res Perspect. 2016. PMID: 27433342 Free PMC article.
-
TRPM2 Channels: A Potential Therapeutic Target in Melanoma?Int J Mol Sci. 2023 Jun 21;24(13):10437. doi: 10.3390/ijms241310437. Int J Mol Sci. 2023. PMID: 37445615 Free PMC article. Review.
-
Insulin secretion stimulated by L-arginine and its metabolite L-ornithine depends on Gα(i2).Am J Physiol Endocrinol Metab. 2014 Nov 1;307(9):E800-12. doi: 10.1152/ajpendo.00337.2014. Epub 2014 Sep 9. Am J Physiol Endocrinol Metab. 2014. PMID: 25205820 Free PMC article.
References
-
- Clapham DE, Julius D, Montell C, Schultz G. International Union of Pharmacology. XLIX. Nomenclature and structure-function relationships of transient receptor potential channels. Pharmacol Rev. 2005;57:427–450. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

