γ-Tocotrienol is a novel inhibitor of constitutive and inducible STAT3 signalling pathway in human hepatocellular carcinoma: potential role as an antiproliferative, pro-apoptotic and chemosensitizing agent
- PMID: 21198544
- PMCID: PMC3087132
- DOI: 10.1111/j.1476-5381.2010.01187.x
γ-Tocotrienol is a novel inhibitor of constitutive and inducible STAT3 signalling pathway in human hepatocellular carcinoma: potential role as an antiproliferative, pro-apoptotic and chemosensitizing agent
Abstract
Background and purpose: Activation of signal transducer and activator of transcription 3 (STAT3) play a critical role in the survival, proliferation, angiogenesis and chemoresistance of tumour cells. Thus, agents that suppress STAT3 phosphorylation have potential as cancer therapies. In the present study, we investigated whether the apoptotic, antiproliferative and chemosensitizing effects of γ-tocotrienol are associated with its ability to suppress STAT3 activation in hepatocellular carcinoma (HCC).
Experimental approach: The effect of γ-tocotrienol on STAT3 activation, associated protein kinases and phosphatase, STAT3-regulated gene products, cellular proliferation and apoptosis in HCC cells was investigated.
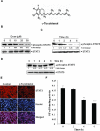
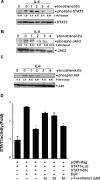
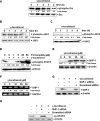
Key results: γ-Tocotrienol inhibited both the constitutive and inducible activation of STAT3 with minimum effect on STAT5. γ-Tocotrienol also inhibited the activation of Src, JAK1 and JAK2 implicated in STAT3 activation. Pervanadate reversed the γ-tocotrienol-induced down-regulation of STAT3, suggesting the involvement of a protein tyrosine phosphatase. Indeed, we found that γ-tocotrienol induced the expression of the tyrosine phosphatase SHP-1 and deletion of the SHP-1 gene by small interfering RNA abolished the ability of γ-tocotrienol to inhibit STAT3 activation. γ-Tocotrienol also down-regulated the expression of STAT3-regulated gene products, including cyclin D1, Bcl-2, Bcl-xL, survivin, Mcl-1 and vascular endothelial growth factor. Finally, γ-tocotrienol inhibited proliferation, induced apoptosis and significantly potentiated the apoptotic effects of chemotherapeutic drugs (paclitaxel and doxorubicin) used for the treatment of HCC.
Conclusions and implications: Overall, these results suggest that γ-tocotrienol is a novel blocker of the STAT3 activation pathway, with a potential role in future therapies for HCC and other cancers.
© 2011 The Authors. British Journal of Pharmacology © 2011 The British Pharmacological Society.
Figures



References
-
- Aggarwal BB, Sethi G, Ahn KS, Sandur SK, Pandey MK, Kunnumakkara AB, et al. Targeting signal-transducer-and-activator-of-transcription-3 for prevention and therapy of cancer: modern target but ancient solution. Ann N Y Acad Sci. 2006;1091:151–169. - PubMed
-
- Aggarwal BB, Vijayalekshmi RV, Sung B. Targeting inflammatory pathways for prevention and therapy of cancer: short-term friend, long-term foe. Clin Cancer Res. 2009b;15:425–430. - PubMed
-
- Ahn KS, Sethi G, Krishnan K, Aggarwal BB. Gamma-tocotrienol inhibits nuclear factor-kappaB signaling pathway through inhibition of receptor-interacting protein and TAK1 leading to suppression of antiapoptotic gene products and potentiation of apoptosis. J Biol Chem. 2007;282:809–820. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

