Endothelial cell-specific deletion of transcription factor FoxM1 increases urethane-induced lung carcinogenesis
- PMID: 21199796
- PMCID: PMC3075588
- DOI: 10.1158/0008-5472.CAN-10-2004
Endothelial cell-specific deletion of transcription factor FoxM1 increases urethane-induced lung carcinogenesis
Abstract
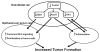
Vascular endothelial cells provide essential support to the tumor microenvironment, but little is known about the transcriptional control of endothelial functions during tumorigenesis. Here we define a critical role for the Forkhead transcription factor FoxM1 in modulating the development of tumor-associated endothelial cells. Pulmonary tumorigenesis induced by urethane administration was compared in mice genetically deleted for FoxM1 in endothelial cells (enFoxm1(-/-) mice). Notably, lung tumor number and size were increased in enFoxm1(-/-) mice. Increased tumorigenesis was associated with increased proliferation of tumor cells and increased expression of c-Myc and cyclin D1. Furthermore, perivascular infiltration by inflammatory cells was elevated and inflammatory cells in BAL fluid were increased. Expression of Flk-1 (vascular endothelial growth factor receptor 2) and FoxF1, known regulators of pulmonary inflammation, was decreased in enFoxm1(-/-) mice. siRNA-mediated knockdown of FoxM1 in endothelial cells reduced Flk-1 and FoxF1 expression, which was driven by direct transcriptional induction by FoxM1 as target genes. Endothelial specific deletion of FoxM1 in vivo or in vitro also decreased expression of Sfrp1 (secreted frizzled-related protein 1), a known inhibitor of canonical Wnt signaling, in a manner that was associated with increased Wnt signaling. Taken together, our results suggest that endothelial-specific expression of FoxM1 limits lung inflammation and canonical Wnt signaling in lung epithelial cells, thereby restricting lung tumorigenesis.
© 2011 AACR.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

