Single particle tracking confirms that multivalent Tat protein transduction domain-induced heparan sulfate proteoglycan cross-linkage activates Rac1 for internalization
- PMID: 21199870
- PMCID: PMC3060510
- DOI: 10.1074/jbc.M110.187450
Single particle tracking confirms that multivalent Tat protein transduction domain-induced heparan sulfate proteoglycan cross-linkage activates Rac1 for internalization
Abstract
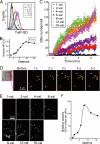
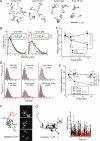
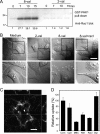
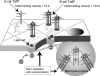
The mechanism by which HIV-1-Tat protein transduction domain (TatP) enters the cell remains unclear because of an insufficient understanding of the initial kinetics of peptide entry. Here, we report the successful visualization and tracking of TatP molecular kinetics on the cell surface with 7-nm spatial precision using quantum dots. Strong cell binding was only observed with a TatP valence of ≥8, whereas monovalent TatP binding was negligible. The requirement of the cell-surface heparan sulfate (HS) chains of HS proteoglycans (HSPGs) for TatP binding and intracellular transport was demonstrated by the enzymatic removal of HS and simultaneous observation of two individual particles. Multivalent TatP induces HSPG cross-linking, recruiting activated Rac1 to adjacent lipid rafts and thereby enhancing the recruitment of TatP/HSPG to actin-associated microdomains and its internalization by macropinocytosis. These findings clarify the initial binding mechanism of TatP to the cell surface and demonstrate the importance of TatP valence for strong surface binding and signal transduction. Our data also shed light on the ability of TatP to exploit the machinery of living cells, using HSPG signaling to activate Rac1 and alter TatP mobility and internalization. This work should guide the future design of TatP-based peptides as therapeutic nanocarriers with efficient transduction.
Figures
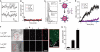
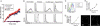

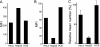
Similar articles
-
Single quantum dot tracking reveals that an individual multivalent HIV-1 Tat protein transduction domain can activate machinery for lateral transport and endocytosis.Mol Cell Biol. 2013 Aug;33(15):3036-49. doi: 10.1128/MCB.01717-12. Epub 2013 Jun 3. Mol Cell Biol. 2013. PMID: 23732912 Free PMC article.
-
Exploring transduction mechanisms of protein transduction domains (PTDs) in living cells utilizing single-quantum dot tracking (SQT) technology.Sensors (Basel). 2012;12(1):549-72. doi: 10.3390/s120100549. Epub 2012 Jan 5. Sensors (Basel). 2012. PMID: 22368485 Free PMC article. Review.
-
ScFv antibody-induced translocation of cell-surface heparan sulfate proteoglycan to endocytic vesicles: evidence for heparan sulfate epitope specificity and role of both syndecan and glypican.J Biol Chem. 2009 Nov 20;284(47):32959-67. doi: 10.1074/jbc.M109.036129. Epub 2009 Sep 25. J Biol Chem. 2009. PMID: 19783663 Free PMC article.
-
HIV-1 Tat and heparan sulfate proteoglycan interaction: a novel mechanism of lymphocyte adhesion and migration across the endothelium.Blood. 2009 Oct 8;114(15):3335-42. doi: 10.1182/blood-2009-01-198945. Epub 2009 Aug 6. Blood. 2009. PMID: 19661268
-
[Mechanism of cellular internalization of various ligands by heparan sulfate proteoglycan].Tanpakushitsu Kakusan Koso. 2008 Sep;53(12 Suppl):1564-9. Tanpakushitsu Kakusan Koso. 2008. PMID: 21089367 Review. Japanese. No abstract available.
Cited by
-
Entry of nanoparticles into cells and tissues: status and challenges.Beilstein J Nanotechnol. 2024 Aug 12;15:1017-1029. doi: 10.3762/bjnano.15.83. eCollection 2024. Beilstein J Nanotechnol. 2024. PMID: 39161463 Free PMC article.
-
Filoviruses utilize glycosaminoglycans for their attachment to target cells.J Virol. 2013 Mar;87(6):3295-304. doi: 10.1128/JVI.01621-12. Epub 2013 Jan 9. J Virol. 2013. PMID: 23302881 Free PMC article.
-
Single quantum dot tracking reveals that an individual multivalent HIV-1 Tat protein transduction domain can activate machinery for lateral transport and endocytosis.Mol Cell Biol. 2013 Aug;33(15):3036-49. doi: 10.1128/MCB.01717-12. Epub 2013 Jun 3. Mol Cell Biol. 2013. PMID: 23732912 Free PMC article.
-
Exploring transduction mechanisms of protein transduction domains (PTDs) in living cells utilizing single-quantum dot tracking (SQT) technology.Sensors (Basel). 2012;12(1):549-72. doi: 10.3390/s120100549. Epub 2012 Jan 5. Sensors (Basel). 2012. PMID: 22368485 Free PMC article. Review.
-
Platelet Activating Factor Receptor Activation Improves siRNA Uptake and RNAi Responses in Well-differentiated Airway Epithelia.Mol Ther Nucleic Acids. 2014 Jul 15;3(7):e175. doi: 10.1038/mtna.2014.26. Mol Ther Nucleic Acids. 2014. PMID: 25025465 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

