Nonthermal dielectric-barrier discharge plasma-induced inactivation involves oxidative DNA damage and membrane lipid peroxidation in Escherichia coli
- PMID: 21199923
- PMCID: PMC3067084
- DOI: 10.1128/AAC.01002-10
Nonthermal dielectric-barrier discharge plasma-induced inactivation involves oxidative DNA damage and membrane lipid peroxidation in Escherichia coli
Abstract
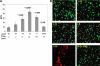
Oxidative stress leads to membrane lipid peroxidation, which yields products causing variable degrees of detrimental oxidative modifications in cells. Reactive oxygen species (ROS) are the key regulators in this process and induce lipid peroxidation in Escherichia coli. Application of nonthermal (cold) plasma is increasingly used for inactivation of surface contaminants. Recently, we reported a successful application of nonthermal plasma, using a floating-electrode dielectric-barrier discharge (FE-DBD) technique for rapid inactivation of bacterial contaminants in normal atmospheric air (S. G. Joshi et al., Am. J. Infect. Control 38:293-301, 2010). In the present report, we demonstrate that FE-DBD plasma-mediated inactivation involves membrane lipid peroxidation in E. coli. Dose-dependent ROS, such as singlet oxygen and hydrogen peroxide-like species generated during plasma-induced oxidative stress, were responsible for membrane lipid peroxidation, and ROS scavengers, such as α-tocopherol (vitamin E), were able to significantly inhibit the extent of lipid peroxidation and oxidative DNA damage. These findings indicate that this is a major mechanism involved in FE-DBD plasma-mediated inactivation of bacteria.
Figures
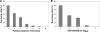

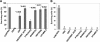
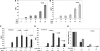
Similar articles
-
Atmospheric Nonthermal Plasma-Treated PBS Inactivates Escherichia coli by Oxidative DNA Damage.PLoS One. 2015 Oct 13;10(10):e0139903. doi: 10.1371/journal.pone.0139903. eCollection 2015. PLoS One. 2015. PMID: 26461113 Free PMC article.
-
Control of methicillin-resistant Staphylococcus aureus in planktonic form and biofilms: a biocidal efficacy study of nonthermal dielectric-barrier discharge plasma.Am J Infect Control. 2010 May;38(4):293-301. doi: 10.1016/j.ajic.2009.11.002. Epub 2010 Jan 20. Am J Infect Control. 2010. PMID: 20085853
-
Mechanisms of Inactivation by High-Voltage Atmospheric Cold Plasma Differ for Escherichia coli and Staphylococcus aureus.Appl Environ Microbiol. 2015 Oct 30;82(2):450-8. doi: 10.1128/AEM.02660-15. Print 2016 Jan 15. Appl Environ Microbiol. 2015. PMID: 26519396 Free PMC article.
-
Functional consequences of oxidative membrane damage.J Membr Biol. 2005 May;205(1):1-16. doi: 10.1007/s00232-005-0753-8. J Membr Biol. 2005. PMID: 16245038 Review.
-
Reactive oxygen species and sperm cells.Reprod Biol Endocrinol. 2004 Mar 23;2:12. doi: 10.1186/1477-7827-2-12. Reprod Biol Endocrinol. 2004. PMID: 15038829 Free PMC article. Review.
Cited by
-
Effects of atmospheric pressure plasmas on isolated and cellular DNA-a review.Int J Mol Sci. 2015 Jan 29;16(2):2971-3016. doi: 10.3390/ijms16022971. Int J Mol Sci. 2015. PMID: 25642755 Free PMC article. Review.
-
Non-thermal Plasma Exposure Rapidly Attenuates Bacterial AHL-Dependent Quorum Sensing and Virulence.Sci Rep. 2016 May 31;6:26320. doi: 10.1038/srep26320. Sci Rep. 2016. PMID: 27242335 Free PMC article.
-
Plasma-activated water: Candidate hand disinfectant for SARS-CoV-2 transmission disruption.Heliyon. 2024 Jul 9;10(15):e34337. doi: 10.1016/j.heliyon.2024.e34337. eCollection 2024 Aug 15. Heliyon. 2024. PMID: 39144986 Free PMC article.
-
Inactivation of Single Strains of Listeria monocytogenes and Salmonella Typhimurium Planktonic Cells Biofilms With Plasma Activated Liquids.Front Microbiol. 2019 Jul 4;10:1539. doi: 10.3389/fmicb.2019.01539. eCollection 2019. Front Microbiol. 2019. PMID: 31333630 Free PMC article.
-
Evaluation of the microbiological safety, quality changes, and genotoxicity of chicken breast treated with flexible thin-layer dielectric barrier discharge plasma.Food Sci Biotechnol. 2016 Aug 31;25(4):1189-1195. doi: 10.1007/s10068-016-0189-1. eCollection 2016. Food Sci Biotechnol. 2016. PMID: 30263393 Free PMC article.
References
-
- Cerutti, P. A. 1985. Prooxidant states and tumor promotion. Science 227:375-381. - PubMed
-
- Flors, C., et al. 2006. Imaging the production of singlet oxygen in vivo using a new fluorescent sensor, Singlet Oxygen Sensor Green. J. Exp. Bot. 57:1725-1734. - PubMed
-
- Flors, C., and S. Nonell. 2006. Light and singlet oxygen in plant defense against pathogens: phototoxic phenalenone phytoalexins. Acc. Chem. Res. 39:293-300. - PubMed
-
- Fridman, A. 2008. Plasma chemistry. Cambridge University Press, New York, NY.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

